
Iron reacts with oxygen as Shown.
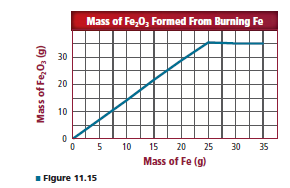
Different amounts of iron were burned in a fixed amount of oxygen. For each mass of iron burned, the mass Of iron(lll) oxide formed was plotted on the graph shown Figure 11.15. Why does the graph level off after 25.0 g of iron is burned? How many moles of oxygen are present in the fixed amount?
Interpretation:
In the given graph, the reason for the level of the graph to be off after 25 g of iron needs to be explained. The number of moles of oxygen present in a fixed amount needs to be determined.
Concept Introduction: Iron reacts with oxygen to produce iron oxide.
Explanation of Solution
A linear increase in the graph suggests Fe2 O3 was produced according to the mass of iron. Up to 25 g of iron is present in fewer amounts. But, after that, oxygen which is present in a fixed amount is not enough to convert the remaining amount of iron to iron oxide. After this point, oxygen acts as a limiting reactant. This is the reason why there is an off on the graph level.
At point 25 g, iron oxygen is present in a sufficient amount. So at this point, the number of moles of oxygen can be calculated.
For that
Step 1: Write a balanced chemical equation
Step 2: Convert the mass of reactant to moles
Step 3: Convert moles of iron to moles of oxygen required using mole ratio
4 mole of Fe require = 3 moles of O2
1 mole of Fe requires
0.447 mole of Fe requires =
Step 4: Convert moles of oxygen to mass
Chapter 11 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Introductory Chemistry (6th Edition)
Chemistry: The Central Science (13th Edition)
CHEMISTRY-TEXT
Organic Chemistry (9th Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





