
The amount of weight percent sulfur.
Answer to Problem 81AAP
The amount of weight percent sulfur is
Explanation of Solution
Show the chemical structure bonding of polyacrylonitrile and polybutadiene as:
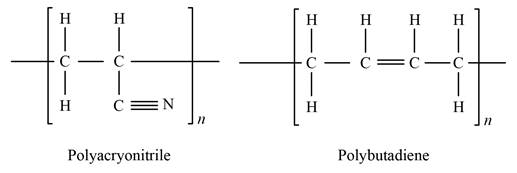
Express the weight per cent of sulfur.
Here, the molar mass of the sulfur is
Calculate the mass of sulfur at 22%.
Here, the number of mole in butadiene is
Calculate the copolymer total mass.
Here, the molar mass of the polybutadlene is
Conclusion:
The average molecular mass of the
The average molecular mass of the polybutadlene is
The average molecular mass of the sulfur is
Substitute
Calculate the fraction of butadlene.
Calculate the number of moles of butadlene using 200 kg of copolymer.
Substitute 3096.3 mol for mole butadlene and
Substitute 21,798 g for
Thus, the amount of weight percent sulfur is
Want to see more full solutions like this?
Chapter 10 Solutions
Foundations of Materials Science and Engineering
- What are the characteristics and mechanical properties of PMMA? Is PMMA a thermoplastic or thermoset polymer?arrow_forwardWhat is the purpose of a plasticizer, and what specific effect on room temperature behavior is likely when a plasticizer is added?arrow_forwardWhat is ceiling temperature in thermodynamics of polymerization? Explain the correlation of ceiling temperature with polymers. Show the equations to support your answer.arrow_forward
- What happens to the stiffness of a polymer as the temperature Tg is exceeded? For what group of polymers is this change the greatest? The smallest?arrow_forward:. Differentiate between Thermoplastics and Thermosets plastics. Explain in detail the uses of these plastics in the world.arrow_forwardExplain why a polymer behaves like a classical Hookean spring when stretched.arrow_forward
- Bakelite is one of the widely ___________________ polymer. Select one: Thermoset Elastomers Composite Thermoplasticarrow_forwardDetermine whether the first part of statement will INCREASE, DECREASE or have NO EFFECT in the second part of the statement. 1. If there is an increase in karat of gold, the strength will ______. 2. If there is an increase in crosslink of a linear polymer, then the degree of amorphousness will _____. 3. If there is an increase in polar groups, then the degree iof polymerization will _____.arrow_forwardFactors that affect the rheological behavior of a polymeric material are (check all that apply)..? Explain and support answer A) Ambient temperature and humidity B) Structural Architecture C) Size and shape of the resin pellets D) Applied stresses and strainsarrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





