
Interpretation: The given diagram needs to be explained using the terms potential energy, kinetic energy, work and heat.
Concept introduction: The ability of doing work or producing heat is the energy. Energy is classified as potential and kinetic energy. The energy due to position or composition is potential energy and the energy because of motion of object is the kinetic energy.
Explanation of Solution
The given diagram is as follows:
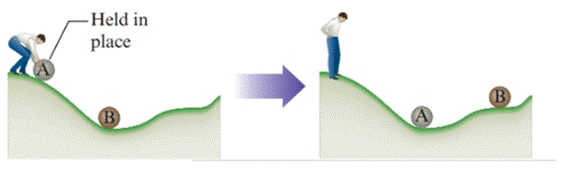
Two balls A and B are present. Ball A present at higher position in the hill has more potential energy than B. When ball A is released, it moves down the hill and strikes ball B, gives the second arrangement (at the right of the picture). In releasing of A, potential energy of A has decreased because its position is lowered. Due to the rolling down of ball, potential energy change of A changes to kinetic energy. The transfer of some part of this energy to ball B takes place which causes the movement of B to higher position. The increase in potential energy of B indicates that work is done on B.
From the diagram it is clear that the final position of B is lower the initial position A that means some energy is lost. Both the balls are at rest in the final position; their motions cannot be attributed to the missing energy. Due to the rolling, transfer of some kinetic energy from ball A to hill occurs as heat. So, the lost energy is transferred to the hill surface as heat.
Chapter 10 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





