
Concept explainers
a.
Interpretation:
The structure of given IUPAC name 3-ethyl-3-methylhexane has to be drawn.
Concept Introduction:
The system which is used to name an organic compound is known as IUPAC Nomenclature. IUPAC are known as International Union of Pure and Applied Chemistry.
There are some rules followed for writing IUPAC Nomenclature for
- The longest chain in the compound has to be selected and it is known as parent name. The parent name represents the number of carbon atom present in the continuous carbon chain. The root name for alkane compound is –ane. The single bonds are formed in alkane compounds.
- The suffix group denotes the
functional group present in a molecule. The prefix group indicates the identity, location and number of substituents attached to the carbon compound. - Then name and number the substituents present in compounds. Then use prefix di-to represent two groups, tri- refers to three groups and so on.
There are some rules followed for writing IUPAC Nomenclature for cycloalkane compounds:
- First identify the number of carbon atoms present in the ring. The numbers of carbon atoms present in ring is used as the parent name. Add suffix name -ane and prefix name cyclo- to the parent name of cycloalkane compound.
- The suffix group denotes the functional group present in a molecule. Then number the substituents present in compound. If there is a single substituent no need to mention number. If the ring has more than one substituent, then start numbering the ring with lowest number to the substituents in alphabetical order. Hence combine the parts to form IUPAC name.
a.
Explanation of Solution
The structure can be derived from the given IUPAC name 3-ethyl-3-methylhexane. First step, look at the end of the name to identify the parent name and suffix name. The longest continuous chain has six carbon atoms and so the parent name is hex-. The root name of alkane compounds are –ane has to be added as suffix name to the parent name. Hence the parent name is given as hexane.
The carbon skeleton of hexane is drawn below,
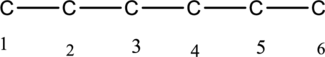
Then number the carbon atoms present in the chain and add the substituents to third carbon. The substituent ethyl groups
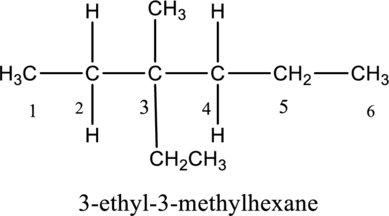
The substituent attached to the carbon skeleton is drawn below,
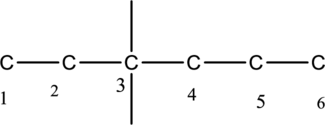
The structure of given IUPAC name 3-ethyl-3-methylhexane is drawn below,
b.
Interpretation
The structure of given IUPAC name 2,2,3,4- tetramethylhexane has to be drawn.
Concept Introduction:
Refer part a.
b.
Explanation of Solution
The structure can be derived from the given IUPAC name 2,2,3,4-tetramethylhexane. First step, look at the end of the name to identify the parent name and suffix name. The longest continuous chain has six carbon atoms and so the parent name is hex-. The root name of alkane compounds are –ane has to be added as suffix name to the parent name. Hence the parent name is given as hexane.
The carbon skeleton of hexane is given below,
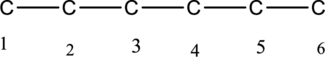
Then number the carbon atoms present in the chain and add the substituents to the least number. The four substituent methyl groups
The substituent is linked to the carbon chain is given below,
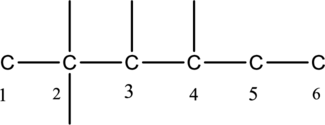
Then name the substituent groups in alphabetical order. Hence the structure of given IUPAC name 2,2,3,4-tetramethylhexane is derived.
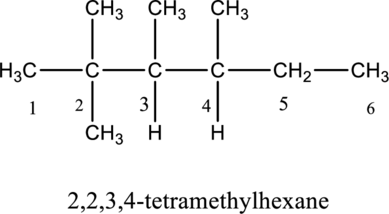
The structure of given IUPAC name 2,2,3,4-tetramethylhexane is drawn below,
c.
Interpretation
The structure of given IUPAC name 4-ethyl-2,2-dimethyloctane has to be drawn.
Concept Introduction:
Refer part a.
c.
Explanation of Solution
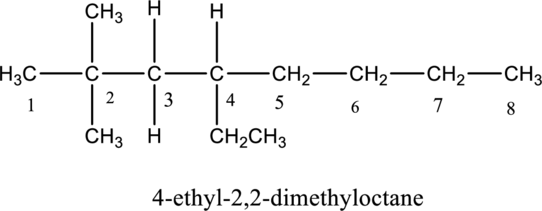
The structure of given IUPAC name 4-ethyl-2,2-dimethyloctane has to be drawn. First step, look at the end of the name to identify the parent name and suffix name. The longest continuous chain has eight carbon atoms and so the parent name is oct-. The root name of alkane compounds are –ane has to be added as suffix name to the parent name. Hence the parent name is given as octane.
The carbon skeleton of octane is drawn below,
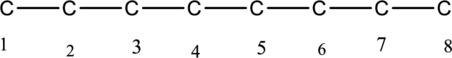
Then number the carbon atoms present in the chain and add the substituents. The two substituent methyl groups
The substituent linked to the carbon chain is drawn below,
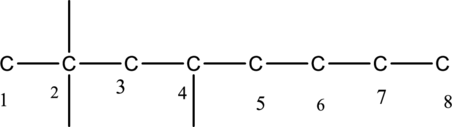
The structure of given compound 4-ethyl-2,2-dimethyloctane is drawn below,
d.
Interpretation
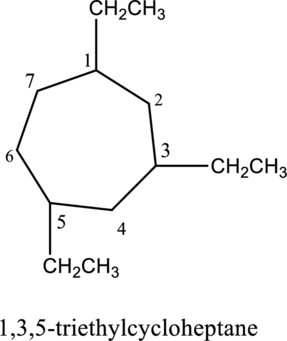
The structure of given IUPAC name 1,3,5-triethylcycloheptane has to be drawn.
Concept Introduction:
Refer part a.
d.
Explanation of Solution
First step, look at the end of the name to identify the parent name and suffix name. The cyclic structure has seven carbon atoms and so the parent name is hept-. The root name of alkane compounds are –ane has to be added as suffix name to the parent name. The prefix name cyclo- represents the cyclic structure. Hence the parent name is given as cycloheptane.
The structure of cycloheptane is drawn below,
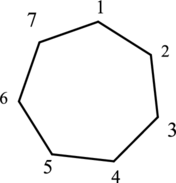
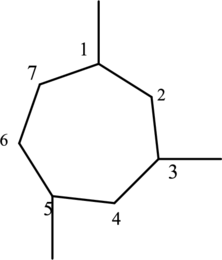
Then number the carbon atoms present in the ring and add the substituents. The three substituent ethyl groups
The substituent attached to the cyclic structure is drawn below,
The structure of given compound 1,3,5-triethylcycloheptane is drawn below,
e.
Interpretation
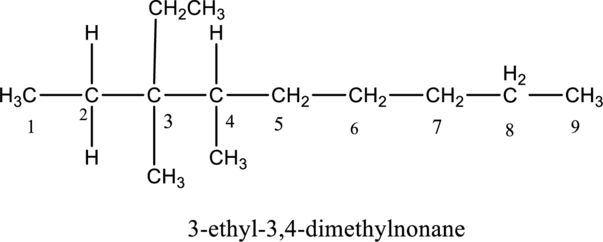
The structure of given IUPAC name 3-ethyl-3,4-dimethylnonane has to be drawn.
Concept Introduction:
Refer part a.
e.
Explanation of Solution
First step, look at the end of the name to identify the parent name and suffix name. The longest continuous chain has nine carbon atoms and so the parent name is non-. The root name of alkane compounds are –ane has to be added as suffix name to the parent name. Hence the parent name is given as nonane.
The carbon skeleton for nonane is drawn below,
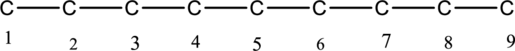
Then number the carbon atoms present in the chain and add the substituents. The two substituent methyl groups
The substituent bonded to carbon chain is drawn below,
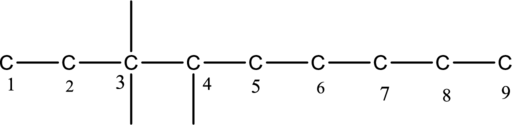
The structure of given compound 3-ethyl-3,4-dimethylnonane is drawn below,
Want to see more full solutions like this?
Chapter 10 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- The correct IUPAC name for the following compound is CH2 H2 H3C. CH3 H2 H3C CH3 a. 2,3,3-trimethyl-1-hexene b. 2,3,3-trimethylhexane c. 4,4,5-trimethylhexane d. 3,4,4-trimethyl-2-hexenearrow_forwardWhat is the IUPAC name of this compound ? A. 3-ethyl-2,7,8-trimethylnonane B. 2,6-diisopropyloctane C. 6-ethyl-2-isopropyl-7-methyloctane D. 7-ethyl-2,3,8-trimethylnonane E. 7-isopropyl-2,3-dimethylnonanearrow_forwardWhat is the IUPAC name of the following compound? O A. 3,4,4-Triethyl-2,2,5-trimethylhexane B. 3-Ethyl-3-isopropyl-4-tertbutylhexane O c. 3,3,4-Triethyl-2,5,5-trimethylhexane D. 3,4-Diethyl-4-isopropyl-2,2- dimethylhexane.arrow_forward
- Draw the structure of each alkane and cycloalkane from the given incorrect name. Then, give the IUPAC name for each compound. a. 7-ethyl-3,6-dimethylnonane b. 4-ethyl-3-isopropylheptane c.3-ethyl-1,4-dimethylcycloheptane d. 1-ethyl-3-methyl-5-isopropylcyclohexanearrow_forward2. What is the IUPAC name of the following compound? CH3 a. 2,4-dimethylhexane b. 2-ethyl-4-methylpentane c. 2,4-methylhexane d. 3,5-dimethylhexane e. 2,4-octane H3C-CH-CH2-CH–CH2-CH3 ČH3arrow_forwardEach of the following IUPAC names is incorrect. Explain why it is incorrect and give the correct IUPAC name. a. 2,2-dimethyl-4-ethylheptane b. 5-ethyl-2-methylhexane c. 2-methyl-2-isopropylheptane d. 1,5-dimethylcyclohexane e. 1-ethyl-2,6-dimethylcycloheptane f. 5,5,6-trimethyloctane g. 3-butyl-2,2-dimethylhexane h. 1,3-dimethylbutanearrow_forward
- 4. The following names are for actual compounds, but the name given are incorrect. Draw out the structures and give the proper IUPAC name. a. 4-ethylpentane b. 2-ethyl-3-methylpentane c. 2,2-diethylheptane d. 2-propylpentane e. 4,4-diethylbutanearrow_forwardExplain why each of the following is an incorrect IUPAC name. Write the correct IUPAC name for the compound. a. 1,3-Dimethylbutane b. 4-Methylpentane c. 2,2-Diethylbutane d. 2-Ethyl-3-methylpentane e. 2-Propylpentane f. 2,2-Diethylheptane g. 2,2-Dimethylcyclopropane h. l-Ethyl-5-methylcyclohexanearrow_forward1. What is the IUPAC name of the compound below? (Please refer to the image attached.) A. Trichloro-1,2,3-propylbenzene B. 2,3,5-Trichlorotoluene C. 1-Propyl-2,3,5-trichlorobenzene D. 1,2,5-Trichloro-3-propylbenzene E. None of the choices 2. Which of the following is a carbonyl containing functional group? I. Aldehyde II. Ketone III. Carboxylic acid IV. Amine A. I,II,III,IV B. I,II,III C. I,II D. I 3. Which of the following functional group contains a hydroxyl group? A. Ester B. Ether C. Aldehyde D. Alcoholarrow_forward
- What is the IUPAC name for the following compound? O A. 2-isopropyl-3-pentyne O B. 4-isopropyl-2-pentyne OC 4,5-dimethyl-2-hexyne O D.2,3-dimethyl-4-hexynearrow_forwardWhich of the following compound has the highest negative heat of combustion? A. 2-methylheptane B.2,4-dimethylhexane C.3-ethyl-2-methylpentane D. 2,3,4 trimethylpentanearrow_forwardO A. 4-ethyl-5-methyloctane O B. 4-methyl-5-ethyloctane OC. 4-methyl-3-propylheptane O D.4-methyl-5-propyloctanearrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning


