
Concept explainers
Interpretation:
The structure of an alkane, an alkene and an
Concept Introduction:
The alkanes are simple organic compounds. These alkane compounds have carbon-carbon single bond and it is not a
The alkene compounds have their functional group as carbon–carbon double bonds. The alkene groups are known as unsaturated compounds. The molecular formula of alkene compounds is given as
Alkyne:
The alkyne compounds have their functional group as carbon-carbon triple bonds. The alkene compounds are known as
Explanation of Solution
The structure of an alkane compound having five carbon atoms has to be drawn. The alkane compounds have carbon-carbon single bond. The first step is to draw the carbon skeleton having five carbon atoms for an alkane compound. Then add hydrogen atom to the carbon in the chain. The carbon belongs to group 4A of periodic table. The carbon has four valence electrons. So the carbon should have four bonds.
The alkane compound having five carbon atoms is drawn below,
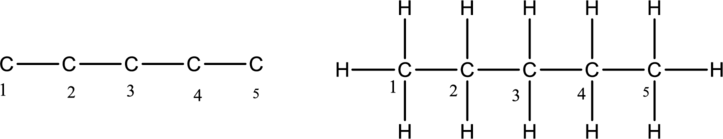
The structure of an alkene compound having five carbon atoms has to be drawn. The alkene compounds have carbon-carbon double bond. The first step is to draw the carbon skeleton having five carbon atoms for an alkene compound. The double bond is bonded between second and third carbon atoms. Then add hydrogen atom to the carbon in the chain. The carbon belongs to group 4A of periodic table. The carbon has four valence electrons. So the carbon should form four bonds.
The alkene compound having five carbon atoms is drawn below,
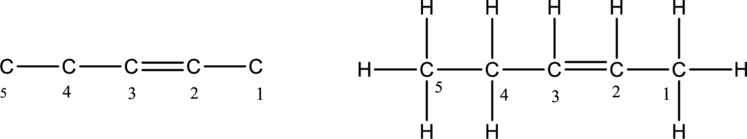
The structure of an alkyne compound having five carbon atoms has to be drawn. The alkyne compounds have carbon-carbon triple bond. The first step is to draw the carbon skeleton having five carbon atoms for an alkyne compound. The triple bond is bonded between second and third carbon atoms. Then add hydrogen atom to the carbon in the chain. The carbon belongs to group 4A of periodic table. The carbon has four valence electrons. So the carbon should form four bonds.
The alkyne compound with five carbon atoms is drawn below,
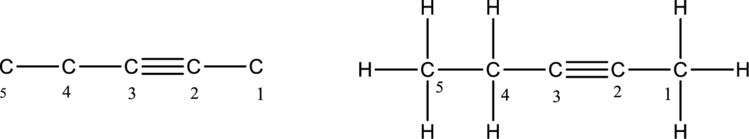
The molecular formula of alkane compound having five carbon atoms is given as,
The molecular formula of alkane compounds can be given as
Given:
The molecular formula of alkene compound having five carbon atoms is given below,
The molecular formula of alkene compounds can be given as
Given:
The molecular formula of alkyne compound having five carbon atoms is given below,
The molecular formula of alkyne compounds are given as
Given:
Want to see more full solutions like this?
Chapter 10 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- How many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forward4.2 What is an alkane?arrow_forwardWhat is meant by the term “unsaturated hydrocarbon”? What structural feature characterizes unsaturated hydrocarbons?arrow_forward
- Complete and balance the following combustion reactions. Assume that each hydrocarbon is converted completely to carbon dioxide and water. (a) Propane + O2 (b) Octane + O2 (c) Cyclohexane + O2 (d) 2-Methylpentane + O2arrow_forwardWrite structures for the three isomers of the aromatic hydrocarbon xylene, C6H4(CH3)2.arrow_forwardWhat is the difference between the hybridization of carbon atoms' valence orbitals in saturated and unsaturated hydrocarbons?arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning  Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





