
Concept explainers
(a)
Interpretation:
Lewis structure for the given molecule is to be completed.
Concept introduction:
Lewis structures involve only valence electrons. When drawing a Lewis structure, the first step is to calculate the total number of valence electrons. For a complete Lewis structure of a molecule, the atoms must complete their normal valency by bond formation and lone pairs of electrons. Maximum number of covalent bonds formed by any neutral atom with maximum number of lone pairs is
| Atom | Number of bond | Number of lone pairs |
| C | 4 | 0 |
| H | 1 | 0 |
| O | 2 | 2 |
| N | 1 | 1 |
| F | 1 | 3 |
Answer to Problem 1.46P
The complete Lewis structure for the given molecule is
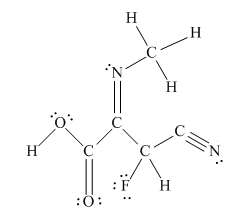
Explanation of Solution
The given structure is
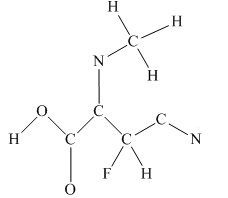
Total valence electron count for the given molecule is
The other oxygen atom has formed only one bond with carbon. This is converted to a double bond and two lone pairs are placed on the oxygen atom so that its octet is complete. A double bond is placed between C and N atom to complete the octet of carbon and a lone pair is placed in nitrogen to complete its octet.
A triple bond is placed between the other C and N to complete the octet of carbon and a lone pair is placed in the nitrogen to complete its octet.

This structure now accounts for all 54 electrons and the octet of each atom, except hydrogen, is complete. The duet for all hydrogens is complete.
The Lewis structure for the given molecule is completed from total valence electron count.
(b)
Interpretation:
Lewis structure for the given molecule is to be completed.
Concept introduction:
Lewis structures involve only valence electrons. When drawing a Lewis structure, the first step is to calculate the total number of valence electrons. For a complete Lewis structure of a molecule, every carbon atom must form four covalent bonds whereas the hydrogen atom forms one bond.
Answer to Problem 1.46P
The complete Lewis structure for the given molecule is
Explanation of Solution
The given structure is
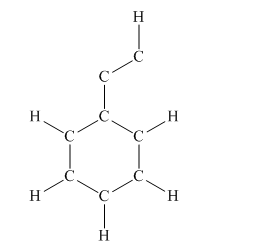
Total valence electron count for the given molecule must be

This structure now accounts for all 38 electrons and the octet of each atom, except hydrogen, is complete. The duet for all hydrogen atoms is complete.
The Lewis structure for the given molecule is completed from total valence electron count.
(c)
Interpretation:
Lewis structure for the given molecule is to be completed.
Concept introduction:
Lewis structures involve only valence electrons. When drawing a Lewis structure, the first step is to calculate the total number of valence electrons. For a complete Lewis structure of a molecule, the atoms must complete their normal valency by bond formation and lone pairs of electrons. Maximum numbers of covalent bonds formed by any neutral atom with maximum number of lone pair are
| Atom | Number of bond | Number of lone pairs |
| C | 4 | 0 |
| H | 1 | 0 |
| O | 2 | 2 |
| N | 1 | 1 |
Answer to Problem 1.46P
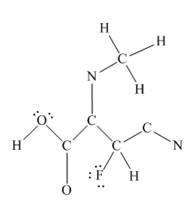
The complete Lewis structure for the given molecule is
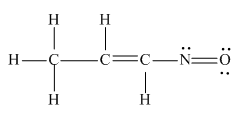
Explanation of Solution
The given structure is
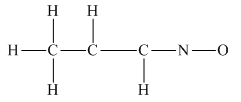
Total valence electron count for the given molecule is
The oxygen atom has formed only one bond with nitrogen. This is converted to a double bond and two lone pairs are placed on the oxygen atom so that its octet is complete. Another lone pair is placed on the nitrogen atom so that its octet is complete.
A double bond is placed between the C atoms attached to one hydrogen each. This completes the octet of both carbon atoms

This structure now accounts for all the 28 electrons, and the octet of each atom, except hydrogen, is complete. The duet for all hydrogen atoms is complete.
The Lewis structure for the given molecule is completed from total valence electron count.
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- For each of the Lewis structures shown below, predict the Electron Geometry, Molecular Geometry and Bond Angle. Lastly, using the same format as shown in the last column of Table 1, draw a sketch (using wedges and dashes to show 3D if needed) of the Molecular Geometry.arrow_forwardDirections: Complete the data table below. It is not necessary to construct any models. Number of bonds & number of lone pairs around central atom Chemical Valence electrons name Formula Lewis structure NCI, N-5 3 bonds Nitrogen CI- 7(3) 20 lone pairs trichloride Si-4 S-6(2) Sis, Silicon disulfide Н-1 C- 4 HCN N-5 C-4 CF,CI, F-7(2) CI-7(2) Ge-4 Germane GeH, H-1(4) C-4 CSF, S-6 F-7(2) Selenium Se-6 dichloride SeCl, CI-7(2) AsF, As-5 F-7(3) arsenic trifluoride C-4(2) Br-7(2) Dibromoacetylene chlorine gas Cl: CI-7(2) dihydrogen monoxide DHMO CH,O C-4 H-1(2) O-6 carbon CF, C-4 tetrafluoride F-7(4)arrow_forwardHow do you know when to draw a solid wedge vs a dashed wedge when drawing 3D bond-line structures? I know that solid-wedge means the atom is pointing towards you and dashed wedge means it's in the back, but how do you know which atoms are in the front as opposed to the back? How can you tell what the configuration will look like in space just by looking at the lewis structure or name?arrow_forward
- Draw the Lewis structures for the following four molecules, being sure to show all steps following the methods covered in class. Structures without work shown will be marked incorrect. Also, one of these molecules has resonance structures – for this compound, make sure to include all resonance structures, indicate formal charges for each atom. SO2 OF2 IF3 NH4+arrow_forwardIn the POF, molecule, the P atom is the central atom. Draw a Lewis diagram of POF, for which all formal charges are equal to zero. How many double bonds are there in the structure that you have drawn? number of double bonds = Draw a Lewis diagram in which the octet rule is satisfied on all atoms. What are the formal charges on the following atoms in the structure that you have drawn? P Based on formal charge, which is the best Lewis structure for the molecule?arrow_forwardUnshared, or lone, electron pairs play an important role in determining the chemical and physical properties of organic compounds. Thus, it is important to know which atoms carry unshared pairs. Use the structural formulas below to determine the number of unshared pairs at each designated atom. Be sure your answers are consistent with the formal charges on the formulas. H3C H₂C a O ao H₂ b b Hc N CH₂ Submit Answer OC₂H5 The number of unshared pairs at atom a is The number of unshared pairs at atom bis The number of unshared pairs at atom cis The number of unshared pairs at atom a is The number of unshared pairs at atom b is The number of unshared pairs at atom cis Retry Entire Group 9 more group attempts remaining V 0 1 2 3 4 Previous Email Instructor Next Save and Exitarrow_forward
- Formal Charge and Resonance 1.48 Each of the following species will be encountered at some point in this text. They all have the same number of electrons binding the same number of atoms and the same arrangement of bonds; they are isoelectronic. Specify which atoms, if any, bear a formal charge in the Lewis formula given and the net charge for each species. (a) :N=N: (e) :C=0: (c) :C=C: (d) :N=0: (b) :C=N:arrow_forwardP2.17: The February 27, 2006 issue of Chemical and Engineering News contains an interesting article on the 100th birthday of Albert Hofmann, the inventor of the hallucinogen LSD. The structure of LSD is shown below. Several minor resonance contributors can be drawn in which the nitrogen atom indicated by an arrow bears a positive formal charge. Indicate atoms where a corresponding negative formal charge could be located in these contributors. H N N. H CH3 D-lysergic acid diethylamine (LSD) Aarrow_forward3. Molecule: CH4 indicate the number of available electrons that are in the molecule. ae = Trial Structure: in the space to the right connect all of the atoms to the central atom and then make each atom follow the octet rule (duet rule for hydrogen). How many electrons are necessary in the trial structure? Circle the correct ne = ne = ae ne ae relationship between ne and ae. Draw the corrected Lewis Structure to the right. Add Later: e- geometry: molecular geom Hybridization:arrow_forward
- Answer each of the following questions for ClF3. (a) total valence electron count. (b) the Lewis structure. (c) the arrangement (or shape) of the electron grouping (includes lone pairs). (d) ideal bond angle. (e) actual molecular geometry (may be the same or different than answer (c). (f) actual bond angles (may use > or< symbols where appropriate).arrow_forwardUnshared, or lone, electron pairs play an important role in determining the chemical and physical properties of organic compounds. Thus, It Is Important to know which atoms carry unshared pairs. Use the structural formulas below to determine the number of unshared pairs at each designated atom. Be sure your answers are consistent with the formal charges on the formulas. H₂C O H₂ b CH₂ C Hc H₂C-CH₂ Submit Answer The number of unshared pairs at atom a is The number of unshared pairs at atom b is The number of unshared pairs at atom c is The number of unshared pairs at atom a is The number of unshared pairs at atom b is The number of unshared pairs at atom c is Retry Entire Group 9 more group attempts remaining Previous Email Instructor Next Save and Exitarrow_forward1. Molecule: CF4 indicate the number of available electrons that electrons ae = are in the molecule. in the space to the right connect all of the atoms to the central atom and then make each atom follow the octet rule Trial Structure: (duet rule for hydrogen). How many electrons are necessary in the trial ne = structure? Circle the correct ne = ae ne ae relationship between ne and ae. Draw the corrected Lewis Structure to the right. Add Later: e- geometry: molecular geOm Hybridization:arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

