
Concept explainers
(a)
Interpretation:
Out of the two distances between two nitrogen atoms, 50 pm or 75 pm, it is to be determined which one represents higher energy.
Concept introduction:
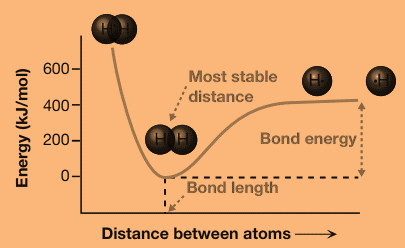
The energy of the molecule depends on the distance between the two atoms. At a certain distance, called the bond length, the energy reaches a minimum. As the distance between the two atoms increases, the energy of the molecule increases and slowly approaches a constant value as the distance increases to infinity. At distances shorter than the bond length, the energy increases very rapidly.
(b)
Interpretation:
Out of the two distances between two nitrogen atoms, 75 pm or 110 pm, it is to be determined which one represents higher energy.
Concept introduction:
The energy of the molecule depends on the distance between the two atoms. At a certain distance, called the bond length, the energy reaches a minimum. As the distance between the two atoms increases, the energy of the molecule increases and slowly approaches a constant value as the distance increases to infinity. At distances shorter than the bond length, the energy increases very rapidly.

(c)
Interpretation:
Out of the two distances between two nitrogen atoms, 110 pm or 150 pm, it is to be determined which one represents higher energy.
Concept introduction:
The energy of the molecule depends on the distance between the two atoms. At a certain distance, called the bond length, the energy reaches a minimum. As the distance between the two atoms increases, the energy of the molecule increases and slowly approaches a constant value as the distance increases to infinity. At distances shorter than the bond length, the energy increases very rapidly.

(d)
Interpretation:
Out of the two distances between two nitrogen atoms, 150 pm or 160 pm, it is to be determined which one represents higher energy.
Concept introduction:
The energy of the molecule depends on the distance between the two atoms. At a certain distance, called the bond length, the energy reaches a minimum. As the distance between the two atoms increases, the energy of the molecule increases and slowly approaches a constant value as the distance increases to infinity. At distances shorter than the bond length, the energy increases very rapidly.

Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- (12) Which one of the following statements about fluo- rometry is FALSE? a) Fluorescence is better detected at 90 from the exci- tation direction. b) Fluorescence is typically shifted to longer wave- length from the excitation wavelength. c) For most fluorescent compounds, radiation is pro- duced by a transitionarrow_forwardDon't used Ai solutionarrow_forwardDon't used Ai solutionarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

