
General, Organic, and Biological Chemistry (3rd Edition)
3rd Edition
ISBN: 9780134042428
Author: Laura D. Frost, S. Todd Deal
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 9.90CP
Naproxen, the active ingredient in Aleve® has the structure shown.The
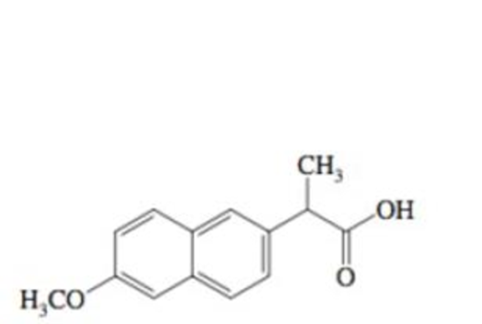
Naproxen, pKa = 7.4
- a. Draw the carboxylate form of Naproxen.
- b. If a person lias diseased tissue with a lower pH, will more or less of the carboxylate form be present?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The pka of butanoic acid is 4.82.
butanoic acid
a. Draw the structure that predominates at a
pH of 1.50.
b. Draw the structure that predominates at a
pH of 9.40.
c. Calculate the ratio of conjugate base to acid
at a pH of 1.50
d. Calculate the ratio of conjugate base to
acid at a pH of 9.40.
At what pH will the concentration of a compound with a pK, 8,4 be 100 tímes greater in ita acidic form than in its basic form?
Express your answer using one decimal place.
pH= 7.9
At what pH will 50% of a compound with a pK, = 7.6 be in its basic form?
Express your answer using one decimal place.
pH =
At what pH will the concentration of a compound with a pK = 4.6 be 10 times greater in its basic form than in its acidic form?
Express your answer using one decimal place.
pH =
Submit
Danunet Anenr
dentify the conjugate base in the reaction of propanoic acid CH3CH3COOH reacting with methyl amine, CH3NH2.
a.
CH3NH2
b.
CH3CH3COOH
c.
CH3NH3+
d.
OH-1
e.
CH3CH3COO-1
Chapter 9 Solutions
General, Organic, and Biological Chemistry (3rd Edition)
Ch. 9 - Prob. 9.1PPCh. 9 - Indicate if each of the following statements is...Ch. 9 - Prob. 9.3PPCh. 9 - Prob. 9.4PPCh. 9 - Prob. 9.5PPCh. 9 - In each of the following equations, identify the...Ch. 9 - Prob. 9.7PPCh. 9 - Prob. 9.8PPCh. 9 - Prob. 9.9PPCh. 9 - Which of the following are strong bases? a....
Ch. 9 - Prob. 9.11PPCh. 9 - Prob. 9.12PPCh. 9 - Prob. 9.13PPCh. 9 - Prob. 9.14PPCh. 9 - Complete and balance the following neutralization...Ch. 9 - Complete and balance the following neutralization...Ch. 9 - Prob. 9.17PPCh. 9 - Complete and balance the following neutralization...Ch. 9 - Prob. 9.19PPCh. 9 - Prob. 9.20PPCh. 9 - Write an equilibrium constant expression lor the...Ch. 9 - Prob. 9.22PPCh. 9 - Prob. 9.23PPCh. 9 - Prob. 9.24PPCh. 9 - Prob. 9.25PPCh. 9 - Sulfur trioxide is produced by reacting sulfur...Ch. 9 - Prob. 9.27PPCh. 9 - Prob. 9.28PPCh. 9 - Prob. 9.29PPCh. 9 - When you exercise, energy is produced by...Ch. 9 - Using Tables 9.1 and 9.6, identity the stronger...Ch. 9 - Using Tables 9.1 and 9.6, identify the stronger...Ch. 9 - Prob. 9.33PPCh. 9 - Identify the acid and base on the reactant side of...Ch. 9 - Prob. 9.35PPCh. 9 - Prob. 9.36PPCh. 9 - Prob. 9.37PPCh. 9 - Write the formula and name of the conjugate acid...Ch. 9 - Complete the following reactions and identify the...Ch. 9 - Complete the following reactions and identify the...Ch. 9 - State if each of the following solutions is...Ch. 9 - State if each of the following solutions is...Ch. 9 - State if each of these following solutions is...Ch. 9 - Slate if each of the following solutions is...Ch. 9 - Calculate the pH of each of the solutions in...Ch. 9 - Calculate the pH of each of the solutions in...Ch. 9 - Calculate the [H3O+] for each of the following...Ch. 9 - Calculate the [H3O+J lor each of the following...Ch. 9 - Prob. 9.49PPCh. 9 - Using Table 9.8, determine the stronger acid from...Ch. 9 - Prob. 9.51PPCh. 9 - Prob. 9.52PPCh. 9 - Prob. 9.53PPCh. 9 - Prob. 9.54PPCh. 9 - Valine has the zwitterion structure shown in the...Ch. 9 - Glycine has the zwitterion structure shown in the...Ch. 9 - Prob. 9.57PPCh. 9 - Prob. 9.58PPCh. 9 - During stress or trauma, a person can start to...Ch. 9 - A person who overdoses on antacids may neutralize...Ch. 9 - Prob. 9.61APCh. 9 - Prob. 9.62APCh. 9 - Prob. 9.63APCh. 9 - What are some ingredients found in antacids? What...Ch. 9 - Prob. 9.65APCh. 9 - Prob. 9.66APCh. 9 - Prob. 9.67APCh. 9 - Prob. 9.68APCh. 9 - For the following reaction, 2HI(g)H2(g)+I2(g) a....Ch. 9 - Prob. 9.70APCh. 9 - Prob. 9.71APCh. 9 - Prob. 9.72APCh. 9 - Prob. 9.73APCh. 9 - Prob. 9.74APCh. 9 - Prob. 9.75APCh. 9 - Prob. 9.76APCh. 9 - Determine the pH for the following solutions....Ch. 9 - Determine the pi! for the following solutions....Ch. 9 - Prob. 9.79APCh. 9 - Prob. 9.80APCh. 9 - Consider the acetic-acid buffer system with acetic...Ch. 9 - Consider the lactic-acid buffer with lactic acid,...Ch. 9 - In blood plasma, pH is maintained by the carbonic...Ch. 9 - Adding a few drops o! a strong add to water will...Ch. 9 - Consider the amino acid valine shown in its...Ch. 9 - Prob. 9.86CPCh. 9 - Prob. 9.87CPCh. 9 - Prob. 9.88CPCh. 9 - Prob. 9.89CPCh. 9 - Naproxen, the active ingredient in Aleve has the...Ch. 9 - To determine the concentration of an unknown weak...Ch. 9 - Explain why the following amino acid cannot exist...Ch. 9 - Prob. 1IA.1QCh. 9 - Prob. 1IA.2QCh. 9 - Prob. 1IA.3QCh. 9 - Prob. 1IA.4QCh. 9 - Prob. 1IA.5QCh. 9 - Provide the a. conjugate base of H2S. _______ b....Ch. 9 - If the lungs fail to expel normal amounts of CO2...Ch. 9 - If the lungs expel CO2 faster than normally...Ch. 9 - Prob. 2IA.3QCh. 9 - Prob. 2IA.4QCh. 9 - Prob. 1ICCh. 9 - Prob. 2ICCh. 9 - Prob. 3IC
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A carboxylic acid has a pKa of 6.00. Which of the following statements is true? O A. There is an equimolar ratio of the acid and its conjugate base when the pH is 6.00. B. Half of the acid will be deprotonated when the pH = 3. O C. Half of the acid will be protonated when the pH = 12. D. The acid will be completely neutralized when the pH = 7.00. %3!arrow_forwardExplain why a pKa is not found for the strong acid, but one will be found for the weak acid.arrow_forwardTrue or false Acidic drugs tend to ionize in basic medium. Basic drugs tend to be absorbed in basic environment, like the duodenum. Acidic drugs are excreted faster if the urine is acidic. Basic drugs remain unionize in a basic environment. Polarity of the drug increases as the number of carbon increases in a compound.arrow_forward
- Given the following information determine which of these acids is the strongest? Ka for boric acid is 7.3 x 10–10 pKa of acetic acid is 4.75 Ka for lactic acid is 8.4 x 10–4 pKa of phenol is 9.89 A acetic acid B formic acid C lactic acid D phenolarrow_forward1. Stomach acid (gastric acid) What is the source of H+ for stomach acid? 2. Acetylcholine >> Into blood Where does the conjugate base of Histamine →→→→ carbonic acid (H₂CO3) go? Stomach acid Acetylcholine → (gastric acid) Gastrin->> ~HCO; > ч нсоз CI- Parietal Cell H,0 + CO, H,CO, < HCO₂ + H+ Proton pump Parietal Cell H,0 + CO, H₂CO3 Carbonic anhydrase HCO₂ + H+ Proton pump Carbonic anhydrase K* ADP+P S K+ ← ADP+P, ATP ATP →→K+ H+ Lumen of gastric pit K+ CI- H* CI H+ CI- H+ Lumen of gastric pit CI- CI- Cl H+ H+ H+ CI- H+ CI H* H+ H* CI- H+ CI- Harrow_forwardNitric acid (HNO3) is a strong acid that completely ionizes when dissolved in water (pKa = -1.30). Calculate the pH of 10 mg/L HNO3. a. Unlike nitric acid, butyric acid (CH3CH2CH2COOH) is a weak acid (pKa = 4.8). Calculate the pH of a solution when 90% of the CH3CH2CH2COOH is dissociated. b. Briefly explain how you can use the pKa values of these two molecules to determine that nitric acid is a strong acid, while butyric acid is a weak acid.arrow_forward
- 18. During acidosis of a protein, shown below, salt bridges are broken, resulting in denaturation. Which choice shows the products of alkalosis? acidosis Н NO salt bridge H-N- Hзо* Н Н H-N- H-N ОН d. Н Н a. Н H-N- Н Н e. ОН H-N- Н c. b.arrow_forwardThe conjugate base of benzoic acid is used as a 72 preservative. Write the equilibrium reaction for this weak acid in aqueous solution. ОН Benzoic acid a. What substances are present at equilibrium? b. At equilibrium, are reactants or products favored, and why? c. Are the concentrations of benzoic acid and benzoate constant or changing at equilibrium? d. What happens to the equilibrium if more hydronium ions are added to the reaction? e. Label the conjugate acid-base pairs.arrow_forward10. Given the pH of 1.5 M chloroacetic acid, CH2CICOOH solution is 1.96. a. Write an equation to show the dissociation of CH2CICOOH in water. b. Calculate the Ka of the acid. c. Calculate the pKb of its conjugate base. d. Which is a stronger base, acetate ion or the conjugate base of CH2CICOOH? (Ka of acetic acid, CH3COOH = 1.8 x 105)arrow_forward
- Name two causes of rancidity in oils What is acid value of oils? How many moles of KOH is required per mole of fat in saponification reactions?arrow_forwardWhich of the following explains why alpha-amino acids, the building blocks of proteins, are nearly 1000 times more acidic than aliphatic carboxylic acids? H3N. R. An a-amino acid An aliphatic acid pK, 5 pKa = 2 O Inductive effects of the aliphatic group destabilize the conjugate base. O Inductive effects of the amino group help stabilize the conjugate base. O Inductive effects of the aliphatic groups help stbilize the conjugate base. O Inductive effects of the amino group destabilize the conjugate base.arrow_forwardPart A: Please calculate pKa of formic acid below: (HCOOH H+ + HCOO-) Part B: What is pH of a solution containing 10 mM formic acid? G°f HCOOH = -372 kJ/molG°f HCOO- = -351 kJ/molG°f H+ = 0 kJ/mol1arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY