
General, Organic, and Biological Chemistry (3rd Edition)
3rd Edition
ISBN: 9780134042428
Author: Laura D. Frost, S. Todd Deal
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 9.85AP
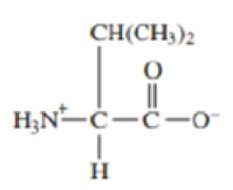
Consider the amino acid valine shown in its zwitterion form The pI of valine is 6.0.
- a. Draw the structure of the predominant form of valine at pH = 10.0.
- b. Draw the structure of thr predominant form of valine at pH = 3.0.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For the amino acid Lysine draw the two predominant species when the pH is 10,You do not need to
draw the entire R group.
What is the pH, along with the concentration of each form of serine, in a 0.020 M
solution of serine?
Calculate [H2Asc], [HAsc-], [Asc2-], and the pH of 0.050 Mascorbic acid H2C6H6O6, H2Asc. (Ka1= 1.0x10-5and Ka2= 5x10-12).
Chapter 9 Solutions
General, Organic, and Biological Chemistry (3rd Edition)
Ch. 9 - Prob. 9.1PPCh. 9 - Indicate if each of the following statements is...Ch. 9 - Prob. 9.3PPCh. 9 - Prob. 9.4PPCh. 9 - Prob. 9.5PPCh. 9 - In each of the following equations, identify the...Ch. 9 - Prob. 9.7PPCh. 9 - Prob. 9.8PPCh. 9 - Prob. 9.9PPCh. 9 - Which of the following are strong bases? a....
Ch. 9 - Prob. 9.11PPCh. 9 - Prob. 9.12PPCh. 9 - Prob. 9.13PPCh. 9 - Prob. 9.14PPCh. 9 - Complete and balance the following neutralization...Ch. 9 - Complete and balance the following neutralization...Ch. 9 - Prob. 9.17PPCh. 9 - Complete and balance the following neutralization...Ch. 9 - Prob. 9.19PPCh. 9 - Prob. 9.20PPCh. 9 - Write an equilibrium constant expression lor the...Ch. 9 - Prob. 9.22PPCh. 9 - Prob. 9.23PPCh. 9 - Prob. 9.24PPCh. 9 - Prob. 9.25PPCh. 9 - Sulfur trioxide is produced by reacting sulfur...Ch. 9 - Prob. 9.27PPCh. 9 - Prob. 9.28PPCh. 9 - Prob. 9.29PPCh. 9 - When you exercise, energy is produced by...Ch. 9 - Using Tables 9.1 and 9.6, identity the stronger...Ch. 9 - Using Tables 9.1 and 9.6, identify the stronger...Ch. 9 - Prob. 9.33PPCh. 9 - Identify the acid and base on the reactant side of...Ch. 9 - Prob. 9.35PPCh. 9 - Prob. 9.36PPCh. 9 - Prob. 9.37PPCh. 9 - Write the formula and name of the conjugate acid...Ch. 9 - Complete the following reactions and identify the...Ch. 9 - Complete the following reactions and identify the...Ch. 9 - State if each of the following solutions is...Ch. 9 - State if each of the following solutions is...Ch. 9 - State if each of these following solutions is...Ch. 9 - Slate if each of the following solutions is...Ch. 9 - Calculate the pH of each of the solutions in...Ch. 9 - Calculate the pH of each of the solutions in...Ch. 9 - Calculate the [H3O+] for each of the following...Ch. 9 - Calculate the [H3O+J lor each of the following...Ch. 9 - Prob. 9.49PPCh. 9 - Using Table 9.8, determine the stronger acid from...Ch. 9 - Prob. 9.51PPCh. 9 - Prob. 9.52PPCh. 9 - Prob. 9.53PPCh. 9 - Prob. 9.54PPCh. 9 - Valine has the zwitterion structure shown in the...Ch. 9 - Glycine has the zwitterion structure shown in the...Ch. 9 - Prob. 9.57PPCh. 9 - Prob. 9.58PPCh. 9 - During stress or trauma, a person can start to...Ch. 9 - A person who overdoses on antacids may neutralize...Ch. 9 - Prob. 9.61APCh. 9 - Prob. 9.62APCh. 9 - Prob. 9.63APCh. 9 - What are some ingredients found in antacids? What...Ch. 9 - Prob. 9.65APCh. 9 - Prob. 9.66APCh. 9 - Prob. 9.67APCh. 9 - Prob. 9.68APCh. 9 - For the following reaction, 2HI(g)H2(g)+I2(g) a....Ch. 9 - Prob. 9.70APCh. 9 - Prob. 9.71APCh. 9 - Prob. 9.72APCh. 9 - Prob. 9.73APCh. 9 - Prob. 9.74APCh. 9 - Prob. 9.75APCh. 9 - Prob. 9.76APCh. 9 - Determine the pH for the following solutions....Ch. 9 - Determine the pi! for the following solutions....Ch. 9 - Prob. 9.79APCh. 9 - Prob. 9.80APCh. 9 - Consider the acetic-acid buffer system with acetic...Ch. 9 - Consider the lactic-acid buffer with lactic acid,...Ch. 9 - In blood plasma, pH is maintained by the carbonic...Ch. 9 - Adding a few drops o! a strong add to water will...Ch. 9 - Consider the amino acid valine shown in its...Ch. 9 - Prob. 9.86CPCh. 9 - Prob. 9.87CPCh. 9 - Prob. 9.88CPCh. 9 - Prob. 9.89CPCh. 9 - Naproxen, the active ingredient in Aleve has the...Ch. 9 - To determine the concentration of an unknown weak...Ch. 9 - Explain why the following amino acid cannot exist...Ch. 9 - Prob. 1IA.1QCh. 9 - Prob. 1IA.2QCh. 9 - Prob. 1IA.3QCh. 9 - Prob. 1IA.4QCh. 9 - Prob. 1IA.5QCh. 9 - Provide the a. conjugate base of H2S. _______ b....Ch. 9 - If the lungs fail to expel normal amounts of CO2...Ch. 9 - If the lungs expel CO2 faster than normally...Ch. 9 - Prob. 2IA.3QCh. 9 - Prob. 2IA.4QCh. 9 - Prob. 1ICCh. 9 - Prob. 2ICCh. 9 - Prob. 3IC
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 3. In the space provided, draw the structure of Glycine (Gly) at the following pH value. Glycine has a pl of 5.97. H. H2N-C-COOH pH = 4.62 %3D H. glycinearrow_forwardConsider the amino acid valine shown in its Zwitterion form. The pI of Vatine is 600. H3-N-CH-2-00 CH-CH3 CH3 a) DRAW THE STRUCTURE OF THE PREDOMINANT FORM OF VALINE AT PH=10.0 DRAW THE STRUCTURE OF THE PREDOMINANT FORM OF VALINE AT H=3.0.arrow_forward1. Exam 4 (Unit 4) Proteins are a type of biological macromolecule, formed from peptide bonds, between what monomer units? amino acids 2. In the space provided, draw the structure of isoleucine at each of the following pH values. Isoleucine has a pl of 6.02. 3 a) pH = 6.02 H H₂N-C-COOH H-C-CH3 SH₂ CH₂ isoleucine b) pH = 5.98arrow_forward
- what does rancidity of oils mean what causes rancidity of oils what is the acid value of oilsarrow_forwardValine (HV) is a diprotic amino acid with Ka1=5.18×10-3 and Ka2=1.91×10-10. Determine the pH of each of the solutions. a) A 0.216 M valine hydrochloride (H2V+CI-) solution. pH= b) A 0.216 M valine (HV) solution. pH= c) A 0.216 M sodium valinate (Na+V-) solution. pH=arrow_forward14 7 12 pK3 10 pH 6 pk2 pl pK1 2 0.5 1.5 2.5 Equivalents of OH 1. What is the net charge of aspartic acid when the pH is 0.10? 2. Draw the pre-dominant form of aspartic acid when the pH is 2.1 3. Draw the zwitterionic form of aspartic acid. Give the approximate pH value wherein the zwitterionic form exist. 8.arrow_forward
- 1. Draw a structural formula for the major ionic form of the amino acid shown below when in aqueous solution at pH 3.0. 2. Draw a structural formula for the major ionic form of the amino acid shown below when in aqueous solution at pH 9.8.arrow_forwardwhat is the ph of 0.29 M of alaninearrow_forwardA dilute (~0.10 M) butanoic acid solution (aka butyric acid) has a pH of 4.6. To the nearest hundredth of a unit, what is the ratio of the acidic form to basic form of the molecules at this pH? (The pKa of butanoic acid is 4.82.)arrow_forward
- Each row represents an experimental “run” (in this case, averaged sample of mango wine). Column A shows the run number; Column B shows the percentage of ethanol; Column C shows the amount of glycerol (in g/L); Column D shows the amount of acid (in g/L); Column E shows the temperature of the run (in °C); and Column F shows the pH of the run. Y represents Ethanol of mango wine. 1. Create a scatterplot of the ethanol data for all 20 runs a. Draw a graphical representation of the model that you chose to use. b. Comparing the model to your data, evaluate its fit in one or two sentences. (Not sure what this question is getting at. I've provided all the information given) c. Replace the generic Yi = β0 + ε with the information you have here. Please help. I have no idea what this even means or where to start. HERE is the attached dataset to start with; a b c d e f Run Ethanol Glycerol Acidity Temp pH 1 4.8 3.5 0.84 24 3.8 2 9.6…arrow_forward3. Draw the two structures in most abundance of the amino acid, cysteine, at a pH of 8.0. What are their respective concentrations in a 50 mM solution? If you had 1.0 L of this solution, how much 3.0 M NaOH would be required to take the pH to 10.4? What two species are now in most abundance (draw the structures)?arrow_forwardIn a highly basic solution, pH = 13, the dominant form of glycine is: a. NH2—CH2—COOH. b. NH2—CH2—COO–. c. NH2—CH3+—COO–. d. NH3+—CH2—COOH. e. NH3+—CH2—COO–.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY