
Microbiology With Diseases By Taxonomy (6th Edition)
6th Edition
ISBN: 9780134832302
Author: Robert W. Bauman Ph.D.
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 9, Problem 1VI
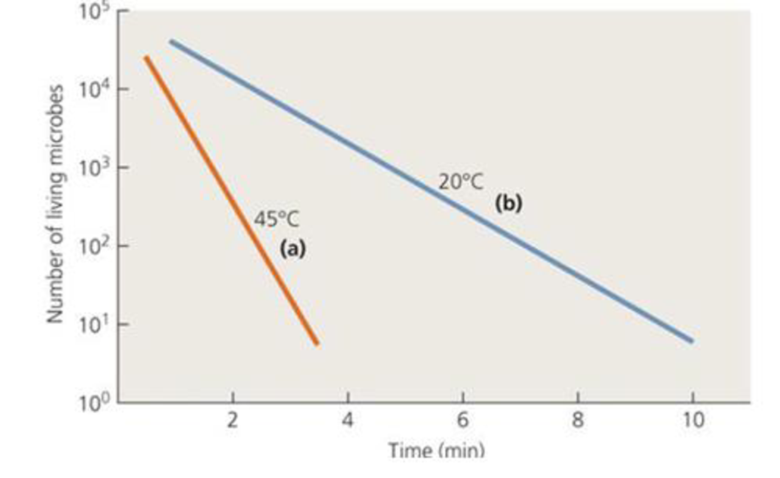
Calculate the decimal reduction time (D) for the two temperatures in the following graph.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
If you measured the rate of reaction at 20°C to be 1.11 x 10-5 M/s when using 0.080 M I1- and 0.040 M S2O82-. Approximately how long will the reaction take if you were to increase the temperature to 30 °C?
Sugar solution is being heated to 83 o C in a jacketed pan made from stainless steel, 1.6 mm thick. Heat is supplied by condensing steam at 200 kPa gauge in the jacket. Calculate the rate of heat transfer if the surface heat transfer coefficients for condensing steam and sugar solution are 12000 J/m2-s-oC and 3000 J/m2-s-oC, respectively. Thermal conductivity for the stainless steel is 21 J/m-s-oC and the surface area of the pan is 1.4 m 2 .
The temperature dependence of the vapor pressure is given by the equation:
ΔΗ
RT
Inp = In A –
Where:
p= vapor pressure
T = temperature
A = pre-exponential constant
AHvap = enthalpy of vaporization
In order to solve for the enthalpy of vaporization, AHvap, you must:
Step One
Add the same expression to each side of the equation to leave the term that includes the variable by itself on the right-hand side of the expression:
(Be sure that the answer field changes from light yellow to dark yellow before releasing your answer)
ΔΗ
RT
+ Inp =
+ In A
Drag and drop your selection from the following list to complete the answer:
1
1
In
In A
- In A
K
Chapter 9 Solutions
Microbiology With Diseases By Taxonomy (6th Edition)
Ch. 9 - Why does milk eventually go bad despite being...Ch. 9 - Why are BSL-4 suits pressurized? Why not just wear...Ch. 9 - Prob. 3TMWCh. 9 - Prob. 4TMWCh. 9 - Prob. 1EDCSCh. 9 - Prob. 2MCCh. 9 - Prob. 3MCCh. 9 - Prob. 4MCCh. 9 - Prob. 5MCCh. 9 - The endospores or which organism can be used as a...
Ch. 9 - A company that manufactures an antimicrobial...Ch. 9 - Prob. 8MCCh. 9 - Prob. 9MCCh. 9 - Prob. 10MCCh. 9 - Prob. 11MCCh. 9 - Which of the following adjectives best describes a...Ch. 9 - Biosafety Level 3 includes ________. a. double...Ch. 9 - Prob. 14MCCh. 9 - Prob. 15MCCh. 9 - Prob. 16MCCh. 9 - Prob. 17MCCh. 9 - Prob. 18MCCh. 9 - Prob. 19MCCh. 9 - Prob. 20MCCh. 9 - Prob. 1SACh. 9 - Compare and contrast four tests that have been...Ch. 9 - Prob. 3SACh. 9 - Prob. 4SACh. 9 - Prob. 5SACh. 9 - Describe five physical methods of microbial...Ch. 9 - Prob. 7SACh. 9 - Prob. 8SACh. 9 - Prob. 9SACh. 9 - Compare and contrast the action of alcohols,...Ch. 9 - Prob. 11SACh. 9 - Why are alcohols more effective in a 70% solution...Ch. 9 - Contrast the structures and actions of soaps and...Ch. 9 - What are some advantages and disadvantages of...Ch. 9 - Prob. 15SACh. 9 - Calculate the decimal reduction time (D) for the...Ch. 9 - Prob. 2VICh. 9 - Prob. 1CTCh. 9 - Is desiccation the only antimicrobial effect...Ch. 9 - Prob. 3CTCh. 9 - Prob. 4CTCh. 9 - Over 1000 people developed severe diarrhea, and at...Ch. 9 - An over-the-counter medicated foot powder contains...Ch. 9 - Prob. 7CTCh. 9 - Prob. 8CTCh. 9 - Explain why quaternary ammonium compounds are not...Ch. 9 - Prob. 10CTCh. 9 - Would you expect Gram-negative bacteria or...Ch. 9 - Where should you place a sterilization indicator...Ch. 9 - Why is liquid water necessary for microbial...Ch. 9 - Prob. 14CTCh. 9 - During the fall 2001 bioterrorist attack in which...Ch. 9 - Prob. 16CTCh. 9 - Prob. 17CT
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Using the data in this table, what is the AG° (in KJ/mol) for the reduction of FAD by water?arrow_forwardCombustion of a fuel sample in a bomb calorimeter increases the temperature of the entire system by 5.10 °C if the calorimeter contains 1700 g of water, but only by 4.00 °C if the calorimeter contains 2200 g of water. What is the heat capacity of the dry bomb calorimeter assembly? Assume that the specific heat capacity of water is 4.18 J g–1 °C–1.arrow_forwardAt what concentration of S(expressed as a multiple of KM) will νo=0.95Vmax?arrow_forward
- Calculate the overall ΔG° (report up to two decimal places) for the net reaction (see attached image). Answer: _____ kcal/mol Note: R = 1.98 x 10 -3 kcal/mol-Karrow_forwardFor the following reaction, 4.91 grams of water are mixed with excess chlorine gas. The reaction yields 12.5 grams of hydrochloric acid.chlorine (g) + water (l) hydrochloric acid (aq) + chloric acid (HClO3) (aq) What is the theoretical yield of hydrochloric acid ? grams What is the percent yield of hydrochloric acid ? %arrow_forwardFor the following results of Thermodynamics of Borax Solubility, the volume of Borax solution titrated by HCI is 8.00 mL. Table 1. Volumes of hydrochloric acid required to titrate a saturated borax solution at varying temperatures. The hydrochloric acid was a solution standardized at 0.2912 M. Borax Volume added (mL) Temp. (°C) 8.00 8.00 8.00 8.00 8.00 HCI Volume (mL) 50.5 33.75 40.7 27.02 30.0 17.95 20.2 13.43 10.3 8.55 Using Thermodynamic formula (R= 8.31 J/K⚫mol) and the above results, (b) What is the y-intercept of the best fitting line for this data? Type your answer...arrow_forward
- For the following results of Thermodynamics of Borax Solubility, the volume of Borax solution titrated by HCI is 8.00 mL. Table 1. Volumes of hydrochloric acid required to titrate a saturated borax solution at varying temperatures. The hydrochloric acid was a solution standardized at 0.2912 M. Borax Volume added (mL) Temp. (°C) 8.00 8.00 8.00 8.00 8.00 HCI Volume (mL) 50.5 33.75 40.7 27.02 30.0 17.95 20.2 13.43 10.3 8.55 Using Thermodynamic formula (R= 8.31 J/K•mol) and the above results, (e) AS° = (J/K⚫mol) Type your answer...arrow_forwardUsing the formula V₁D₁ = V₂D2, what volume of a 1/100 (10-2) dilution would you need to produce 100 mL of a 1/10000 (10-4) dilution? (Note: provide your answer in decimal format to three decimal places.)arrow_forwardPlot a line weaver-burk graph for Km=6.30mM and Vmax=360uM/min when an experiment has 5 tubes with concentrations of substrates of 1.0mM, 10mM, 50mM, and 100mM.arrow_forward
- Calculate the Keq (report up to two decimal places and do not use scientific notation) for the net reaction at 298.15K. (see attached image) Note: R = 1.98 x 10 -3 kcal/mol-Karrow_forwardA mixture containing only KCl and NaBr is analyzed by the Mohr Method. A 0.3172-g sample is dissolved in 50 mL water and titrated to the Ag2CrO4 endpoint, requiring 36.85 mL of 0.1120 M AgNO3. A blank titration requires 0.71 mL of titrant to reach the same endpoint. Report the % (w/w) KCl and NaBr in the sample. [Ans . 84.41 % (w /w); 17.59 % (w /w)]arrow_forwardAt 298 K the standard enthalpy of combustion of sucrose is −5797 kJ mol−1 and the standard Gibbs energy of the reaction is −6333 kJ mol−1. Estimate the additional non-expansion work that may be obtained by raising the temperature to blood temperature, 37 °C.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you


Haematology - Red Blood Cell Life Cycle; Author: Armando Hasudungan;https://www.youtube.com/watch?v=cATQFej6oAc;License: Standard youtube license