
Concept explainers
(a)
Interpretation: The more reactive alkene towards the acid-catalyzed hydration is to be interpreted.
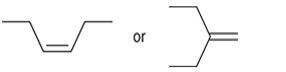
Concept introduction:
(b)
Interpretation: The more reactive alkene from 2-methyl-2-butene and 3-methyl-1-butene toward the acid-catalyzed hydration is to be interpreted.
Concept introduction:
Alkenes are unsaturated hydrocarbons with at least one double bond between the carbon atoms. The presence of pi bonds in these molecules makes them more reactive compared to saturated hydrocarbons; alkanes. The stability of alkenes depends on the substituted groups.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
- 1. what are the differences between acyclic and cyclic forms in terms of their physical properties? 2. what is the role of vinegar in liquid-liquid extraction?arrow_forwardWrite the major carbon containing product. Write optically active or racemic and if the reaction is E1, E2, SN1 or SN2arrow_forwarda) Write out the 3-step arrow pushing mechanism showing how 1-pentene is hydrated to make 2-pentanol. b) Draw the other 2 alkenes(don’t forget cis/trans isomers!) that could also be hydrated to make 2-pentanol. Briefly explain why1-pentene is the best choice.arrow_forward
- Explain why the addition of HBr to alkenes A and C is regioselective, forming addition products B and D, respectively.arrow_forwardExplain why the addition of HBr to alkenes A and C is regioselective,forming addition products B and D, respectively.arrow_forwardIllustrate Comparing Methods of Alkene Synthesis ?arrow_forward
- What is major product (structure and Name) according to Zaitsev’s rule for the dehydration of 3-methyl-2-hexanol?arrow_forwardName the major product(s) formed on mononitration of (a) 1,3-dinitrobenzene, and (b) 1-methyl-4-nitrobenzene. Please also provide the the name in words. not just the structure.arrow_forwardThe careful choice of an appropriate solvent will play a major role in whether organic reactions will be successful. For example, reagents such as CH3CH2Li and NaNH2 will be incompatible with solvents such as water and ethanol. Why is this?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

