
Concept explainers
Interpretation:
The diatomic gas which has shortest bond between its two atoms is to be predicted.
Concept introduction:
The energy required to break a bond is termed as
Answer to Problem 7STP
The
Explanation of Solution
Reason for correct option: It is known that the bond length of bond decreases as the number of bonds between atoms increases. For example, single bond has longest bond length and triple bond has shortest bond length. The bond length also depends on the bond dissociation energies. The longest bond has lowest bond energy and the shortest bond has highest bond energy.
The given table is shown below.
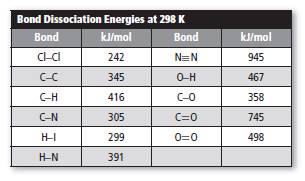
The given diatomic gas is
Among the given diatomic gases,
Reason for incorrect option: The bond dissociation energy of
The bond dissociation energy of
The bond dissociation energy of
Chapter 8 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Organic Chemistry (9th Edition)
Introductory Chemistry (6th Edition)
CHEMISTRY-TEXT
Chemistry: Structure and Properties (2nd Edition)
Chemistry: A Molecular Approach
General, Organic, and Biological Chemistry (3rd Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





