
(a)
INTREPRETATION:
The products obtained from addition of
CONCEPT INTRODUCTION:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Oxidizing Reagents: The chemical agents used to add oxygen or remove hydrogen which finally reduced on oxidizing the other compound.
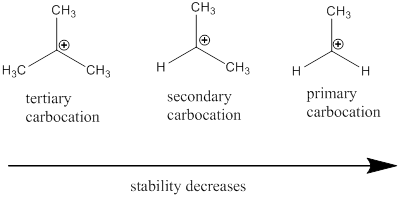
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
(b)
INTREPRETATION:
The mechanism for addition of
CONCEPT INTRODUCTION:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Chemical reaction involves bond making and breaking of two or more reactants in order to attain products from the reactants.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of alkenes when two substituents are placed on same side of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Oxidizing Reagents: The chemical agents used to add oxygen or remove hydrogen which finally reduced on oxidizing the other compound.
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:

Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry (8th Edition)
- 5. Consider the following reaction. H2O2 (excess) NaOH BH3, THE Intermediate A a. Predict the structure of Intermediate A (hint: it is a cyclic compound), then draw a mechanism for its formation. b. Predict the structure of product(s) B.arrow_forwardIdentify the missing reagent(s), reactant(s) or final product(s). Some transformations may require more than one step. A. B. C. MeO₂C CO₂Me Br₂, H₂O 70 °C CN CN Br ? Br D. E. in ? ? LIAIHarrow_forward2. Consijder the reactiojn scheme below a. Draw the major product of this reaction. Show clearly (using wedge-and-dash lines) all stereoisomers formed under these reaction conditions. 1. BH3:THF 2. H2O2, NaOH b. What is the stereochemical relationship between the different stereoisomer products you drew in part 2a.?arrow_forward
- 6. Provide the reagents or the product for the following reactions. Br a b. HO он C. PB13arrow_forwardWhat are the major products of the following reactions? Include stereochemistry. a. 1. Hg(OAc)₂, CH₂OH 2. NaBH4, H₂O, EtOH b. 1. Hg (OAc)₂, (CH3)₂CHOH 2. NaB H₁, H₂O, EtOHarrow_forwardThe following reaction can potentially produce three different cyclized products. 1. NaOMe, MeOH MeO cyclization products 2. acidic workup Provide the structures of the three cyclization products. Label the products as A, B, and C. Circle the major product. Five and six membered rings form preferentially over smaller and larger'rings.arrow_forward
- 4G 08:57 The following reaction schemes have flaws as presented. Which of the following statement is the best description of the flaws in these schemes? CI 1HND, HS0. -CH3 2. CH,a, ACI, 3. Snč, H,o 4. NaOH, HO NH2 OA Step 1 (nitration) of the reaction cannot occur after chlorination OB After nitration, the alkylation reaction cannot occur Oc The reduction of NO2 by SnCl2, H3o* to NH2 should be the last step OD. All reaction steps cannot occur since the ring is deactivated Add a caption... > Status (Custom)arrow_forwardThese reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O, HgSO4 choices:A,DB,CA,B,CA,B,C,Darrow_forwardWhich is the MAJOR product of the following reaction? Et 1) BH3:THF 2) H2O2, NaOH Which of the following best describes a key step in the mechanism for the reaction below? HO ... CH3 -CH3 dihydroxylation + en H3C- H3C- HO. electrophilic addition reaction to form a carbocation intermediate B nucleophilic attack by an alkene to form a cyclic (epoxide) intermediate elimination reaction by abstraction of a beta-hydrogen D free-radical substitution at the carbonyl carbon Which alkene will produce the HIGHEST yield of the alkyl halide below? Br. alkene HBr |arrow_forward
- Draw the product formed from cyclohex-2-enone in each of three reactions. Reaction 1 Reaction 2 Reaction 1 1. NaCN 1. (CH3)2CuLi Reaction 2 product product 2. H3O*, H20 2. CH3CH21 A 1. EtO,CCH,CO¿Et NaOEt, HOET 2. Hао", Н-0, д Reaction 3 Reaction 3 productarrow_forward4. Which of the following reactions is NOT a method of preparation of alkylhalide? A. addition of halogens to alkenes B. replacement of the -OH in alcohol by a halide C. dehydrohalogenation of hydrocarbon D. addition of hydrogen halide to alkene 5. In SN₂ reaction of alkylhalide(substrate) with NaOH(nucleophile), the rate of the reaction is tripled if the concentration of: A. Substrate and nucleophile are both doubled B. Nucleophile is decreased by 1/3 and substrate doubled C. Substrate is tripled and nucleophile doubled D. Nucleophile is tripled 6. Arrange the following alkylhalide in order of decreasing reactivity in an SN₁ reaction: I. CH3CH(Br)CH₂CH3 A. I>II>III>IV II. CH3CH(C1)CH₂CH3 III. CICH₂CH₂CH₂CH3 IV. (CH3)2C(Br)CHCH3 D. IV>II>I>III B. IV>III>II>I C. IV>I>II>IIIarrow_forwardQ5. Regarding the following reaction: Clas na loon CH3 OOH A a. What is the major product? Explain why. b. If the product is compound B, provide a two-step synthesis to give compound B from reactant A. Give the reaction mechanism of the first step. B CH3arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





