
Concept explainers
(a)
Interpretation:
Using curved arrows indicate the flow of electrons for the given mechanism.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of
Acid Catalyzed Hydration Reaction: The reaction involves breaking of phi bonds between carbon-carbon multiple bonds and addition of alcohol to more substituted position of carbon in the molecule.
First step is the acid donates proton to the alkene which leads to the formation of more stable carbo cation.
Then, the water is added to the given alkene through acid catalyzed reaction where the water gets added to the carbo cation finally, the removal of one proton from oxonium ion (oxygen with one positive charge) using water results in the formation of product.
(b)
Interpretation:
The reaction coordinate diagram for the given reaction should be determined.
Concept introduction:
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of alkenes when two substituents are placed on same side of
Acid Catalyzed Hydration Reaction: The reaction involves breaking of phi bonds between carbon-carbon multiple bonds and addition of alcohol to more substituted position of carbon in the molecule.
First step is the acid donates proton to the alkene which leads to the formation of more stable carbo cation.
Then, the water is added to the given alkene through acid catalyzed reaction where the water gets added to the carbo cation finally, the removal of one proton from oxonium ion (oxygen with one positive charge) using water results in the formation of product.
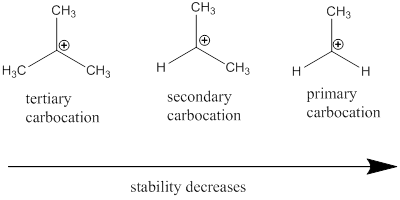
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry (8th Edition)
- please help with this question. thank you. identify the starting material to make each acetal.arrow_forwardDraw the reaction scheme. Use one arrow direct from starting materials to products.arrow_forward1. Provide the reagent(s) needed to convert the given functional group into a new product. Use the Synthesis Map to help navigate between functional groups. a. Transform a primary alcohol into... An alkoxide An aldehyde An alkyl chloride An alkene A carboxylic acid b. Transform an alkane into... An alkyl halide c. Transform a secondary alkyl bromide into... An alcohol An organolithium reagent An alkene d. Transform chloromethane into... A Grignard reagent A symmetric ether (dimethyl ether)arrow_forward
- 8. Draw a reaction coordinate diagram for ... an endergonic reaction *** *** Free energy Free energy Free energy Progress of the reaction a three-step reaction Progress of the reaction ... a reaction that proceeds through two intermediates Progress of the reaction Free energy Progress of the reaction a reaction that proceeds through one transition state *** Progress of the reaction a two-step reaction, in which the *** a two-step reaction, in which the first second step is the rate-determining step step has the highest energy of activation Free energy Free energy Progress of the reactionarrow_forwardDraw curved arrows for the following reaction step.arrow_forwardExplain why two substitution products are formed in the attached reaction.arrow_forward
- Complete the next reactionarrow_forwardFor alkylhalides, elimination reaction is much favored than substitution reaction when A. Temperature is relatively high. B. Temperature is relatively low. C. Base is relatively weak D. Pressure is relatively high.arrow_forwardDraw a stepwise, detailed mechanism for the attached reaction. Usecurved arrows to show the movement of electrons.arrow_forward
- Please indicate an arrow push for each reaction.arrow_forwardClick the "draw structure" button to activate the drawing utility. Draw the major product formed when the following alkene is treated with HCI. draw structure ...arrow_forwarddraw an energy diagram that follows the criteria belowarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
