
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5.6, Problem 5.14P
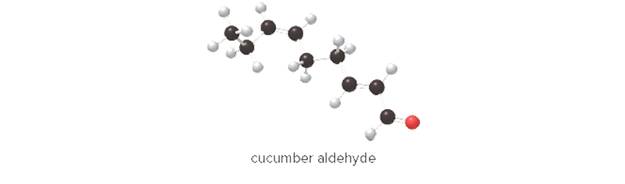
The unmistakable odor of a freshly cut cucumber is due to cucumber
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
. (a) Write the molecular formula of acetylsalicylic acid (see Fig. 7.34a).(b) An aspirin tablet contains 325 mg acetylsalicylic acid. Calculate the number of moles of that compound in the tablet.
3.39 (a) Which of the following molecules can hydrogen bond to another molecule like itself? (b) Which of the following molecules
eán hydrogen bond to water?
OH
СНО
NH
N.
A
F.31 Provide the IUPAC name for each of the following molecules.
(a)
0 (b)
OH
0
(c)
(d) 0
" "I
N
CI
N
NO₂
Chapter 5 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 5.1 - Use the molecular art to identify the process as a...Ch. 5.1 - Use the molecular art to identify the process as a...Ch. 5.1 - Label the reactants and products, and indicate how...Ch. 5.1 - One term in a balanced chemical equation contained...Ch. 5.2 - Write a balanced equation for each reaction....Ch. 5.2 - Write a balanced equation for the following...Ch. 5.2 - Write a balanced equation for the reaction of...Ch. 5.2 - Consider the decomposition of hydrogen peroxide...Ch. 5.2 - Balance each chemical equation. Al+H2SO4Al2(...Ch. 5.3 - Write a balanced equation for the reaction...
Ch. 5.3 - Classify each reaction as a combination or...Ch. 5.3 - Classify each reaction as a single replacement or...Ch. 5.3 - Classify each reaction as a combination,...Ch. 5.3 - Fill in the needed reactants or products for each...Ch. 5.4 - Identify the species that is oxidized and the...Ch. 5.4 - Identify the species that is oxidized and the...Ch. 5.4 - (a) Write a balanced equation for the reaction...Ch. 5.4 - Prob. 5.10PCh. 5.5 - How many items are contained in one mole of (a)...Ch. 5.5 - How many carbon atoms are contained in each of the...Ch. 5.5 - How many molecules are contained in each of the...Ch. 5.5 - How many moles of water contain each of the...Ch. 5.6 - Calculate the formula weight of each ionic...Ch. 5.6 - The unmistakable odor of a freshly cut cucumber is...Ch. 5.6 - Prob. 5.11PPCh. 5.6 - Calculate the number of grams contained in each of...Ch. 5.6 - How many moles are contained in each of the...Ch. 5.6 - How many moles are contained in a 1,000.-mg dose...Ch. 5.6 - How many molecules are contained in two 500.-mg...Ch. 5.7 - Use the balanced equation for the reaction of N2...Ch. 5.7 - Use the balanced equation in Sample Problem 5.15...Ch. 5.8 - Prob. 5.16PPCh. 5.8 - Using the balanced equation for the combustion of...Ch. 5.8 - Prob. 5.17PPCh. 5.8 - Use the balanced equation, N2+O22NO, to answer the...Ch. 5.9 - What is the percent yield of X in a reaction that...Ch. 5.9 - Using the chemical equation in Sample Problem...Ch. 5.9 - Using the equation in Sample Problem 5.20, answer...Ch. 5.9 - The synthetic antiviral drug Tamiflu, currently...Ch. 5.10 - Consider the reaction of hydrogen and nitrogen to...Ch. 5.10 - Using the balanced equation for the reaction of H2...Ch. 5.10 - Using the balanced equation, 3H2(g)+N2(g)2NH3(g),...Ch. 5.10 - Using Sample Problem 5.21 as a guide, complete the...Ch. 5.10 - Using the balanced equation, N2(g)+O2(g)2NO(g),...Ch. 5.10 - Prob. 5.24PPCh. 5 - Prob. 23PCh. 5 - Prob. 24PCh. 5 - How many atoms of each element are drawn on each...Ch. 5 - How many atoms of each element are drawn on each...Ch. 5 - Use the molecular art to write a balanced equation...Ch. 5 - Use the molecular art to write a balanced equation...Ch. 5 - Use the molecular art to write a balanced equation...Ch. 5 - Some coal is high in sulfur (S) content, and when...Ch. 5 - Balance each equation. a....Ch. 5 - Balance each equation. a....Ch. 5 - Prob. 33PCh. 5 - Prob. 34PCh. 5 - Prob. 35PCh. 5 - Prob. 36PCh. 5 - For the reaction depicted in the molecular art:...Ch. 5 - Answer the questions in Problem 5.37 for the...Ch. 5 - Prob. 39PCh. 5 - Prob. 40PCh. 5 - Classify the reaction depicted in the molecular...Ch. 5 - Classify the reaction depicted in the molecular...Ch. 5 - Classify each reaction as combination,...Ch. 5 - Classify each reaction as combination,...Ch. 5 - Fill in the needed reactant or product for each of...Ch. 5 - Fill in the needed reactant or product for each of...Ch. 5 - Identify the species that is oxidized and the...Ch. 5 - Identify the species that is oxidized and the...Ch. 5 - Prob. 49PCh. 5 - Rechargeablenickel-cadmium batteries are used in...Ch. 5 - The reaction of hydrogen (H2) with acetylene...Ch. 5 - Prob. 52PCh. 5 - Calculate the formula weight and molar mass of...Ch. 5 - Calculate the formula weight and molar mass of...Ch. 5 - L-Dopa is a drug used to treat Parkinson’s...Ch. 5 - Niacin, vitamin B3, is found in soybeans, which...Ch. 5 - Which quantity has the greater mass? 1 mol of Fe...Ch. 5 - Prob. 58PCh. 5 - Mescaline is a hallucinogen in peyote, a cactus...Ch. 5 - Prob. 60PCh. 5 - How many grams are contained in 5.00 mol of each...Ch. 5 - How many grams are contained in 0.50 mol of each...Ch. 5 - A bottle of the pain reliever ibuprofen (C13H18O2,...Ch. 5 - One dose of Maalox contains 500. mg each of...Ch. 5 - How many moles are contained in each number of...Ch. 5 - How many moles are contained in each number of...Ch. 5 - How many molecules of butane (C4H10) are contained...Ch. 5 - Prob. 68PCh. 5 - The average nicotine (C10H14N2, molar mass 162.3...Ch. 5 - How many moles of sucrose...Ch. 5 - What is the mass in grams of each quantity of...Ch. 5 - What is the mass in grams of each quantity of...Ch. 5 - Using the balanced equation for the combustion of...Ch. 5 - Sodium metal (Na) reacts violently when added to...Ch. 5 - Prob. 75PCh. 5 - Using the balanced equation for the reaction of Na...Ch. 5 - What is the percent yield of B in a reaction that...Ch. 5 - What is the percent yield of B in a reaction that...Ch. 5 - The reaction of methane (CH4) with Cl2forms...Ch. 5 - Methanol (CH4O), which is used as a fuel in...Ch. 5 - Consider the given reaction mixture that contains...Ch. 5 - Consider the reaction of A2 and B2 to form A2B,...Ch. 5 - Prob. 83PCh. 5 - Consider the reaction with the balanced equation,...Ch. 5 - Using the balanced equation, 2NO+O22NO2, determine...Ch. 5 - Prob. 86PCh. 5 - Prob. 87PCh. 5 - Completer the followin table using the given...Ch. 5 - The local anesthetic ethyl chloride...Ch. 5 - The solvent dischloromethane...Ch. 5 - Answer the following questions about the...Ch. 5 - Answer the following questions about diethyl ether...Ch. 5 - Prob. 93PCh. 5 - Prob. 94PCh. 5 - Prob. 95PCh. 5 - Prob. 96PCh. 5 - TCDD, also called dioxin...Ch. 5 - Prob. 98CP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
2. Why shouldn’t you work in a laboratory by yourself?
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (13th Edition)
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
Basic Chemistry (5th Edition)
22.102 Write the structures of the cis and tram isomers, if any, for the following compounds:
Chemistry: The Molecular Nature of Matter
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In 1934, Edward A. Doisy of Washington University extracted 3000 lb of hog ovaries to isolate a few milligrams of pure estradiol, a potent female hormone. Doisy burned 5.00 mg of this precious sample in oxygen and found that 14.54 mg of CO2 and 3.97 mg of H2O were generated.(a) Determine the empirical formula of estradiol.(b) The molecular weight of estradiol was later determined to be 272. Determine the molecular formula of estradiolarrow_forward(i) Write the name and draw the structure of a saturated hydrocarbon with four carbon atoms.arrow_forward(1) What is the valence electron configuration for the carbon atom? (2) What is the valence electron configuration for the carbon atom?arrow_forward
- Identify the intermolecular forces (dipole–dipole, London dispersion, ydrogen bonding) that influence the properties of the following compounds: (a) Ethane, CH3CH3(b) Ethanol, CH3CH2OH(c) Chloroethane, CH3CH2Clarrow_forward(a) What is a functional group? (b) What functional groupcharacterizes an alcohol? (c) Write a structural formula for1-pentanol, the alcohol derived from pentane by making asubstitution on one of the carbon atoms.arrow_forward(b) alcohol OH 1. H3PO4 2. Os04 3. NaHSO3 در ? ChemDoodlearrow_forward
- Following is the structural formula of acetylsalicylic acid, better known by its common name aspirin. (a) Name the two oxygen-containing functional groups in aspirin. (b) What is the molecular formula of aspirin?arrow_forward(a) When the metallic element sodium combines with the nonmetallic element bromine, Br2(l), how can you determine the chemical formula of the product? How do you know whether the product is a solid, liquid, or gas at room temperature? Write the balanced chemical equation for the reaction. (b) When a hydrocarbon burns in air, what reactant besides the hydrocarbon is involved in the reaction? What products are formed? Write a balanced chemical equation for the combustion of benzene C6H6(l), in air.arrow_forwardAn alkane, P, has the molecular formula, C,H.. An alkene, Q, has the molecular formula, C H,. (a) Name P and Q ánd write their full structural formulae. (b) State two differences between P and Q in terms of their structures. x'arrow_forward
- (a) Give the molecular formula for each of the following. NEC "ool (b) Br OH (c)arrow_forwardCream of tartar is a white powder sometimes used in baking. (It's what separates a tangy, chewy snickerdoodle from an ordinary cinnamon-coated sugar cookie. The acid in the cream of tartar gives snickerdoodles their distinctive tangy flavor, and the chew happens because cream of tartar prevents sugar in the cookie dough from crystalizing into crunchiness. Allrecipies.com) Cream of tartar (KHC4H4O6) is the conjugate base salt of tartaric acid (shown to the right). The Ka for another similar acid is 8.7×10-3. What is the standard Gibb's free energy (in kJ/mol) for the dissociation of that other acid?arrow_forward(d) H₂C=CH₂ NaOH(aq), CHCl3, A CI CIarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY