
(a)
Interpretation: Theacid base reaction of benzoic acid with sodium hydroxide needs to be explained.
Concept Introduction:Bronsted and Lowry purposed the Bronsted-Lowry acid-base theory. It states that acid can give
(b)
Interpretation: The acid, base, conjugated acid and conjugated base in the reaction of benzoic acid with sodium hydroxide needs to be determined.
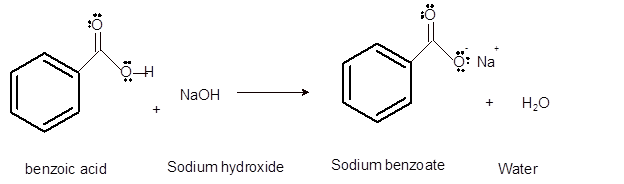
Concept Introduction:Bronsted and Lowry purposed the Bronsted-Lowry acid-base theory. It states that acid can give
(c)
Interpretation: The solubility of benzoic acid and its conjugated base in diethyl ether and water needs to be explained.
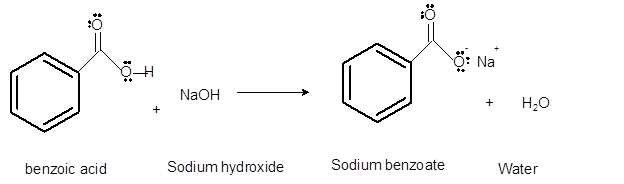
Concept Introduction:Bronsted and Lowry purposed the Bronsted-Lowry acid-base theory. It states that acid can give
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Write the reaction between ammonia, NH3, and hydrosulfuric acid, H2S. NH3 + + In this reaction completes the reactant side while and complete the products side. а. NH3 b. NH4+1 с. HS-1 d. H2S е. Н20 f. H30+1 9. ОН-1arrow_forwardFor the Reaction: NH3+ H2O = NH4+ OH- When adding NH3+ phenolphthalien+ HCl What stress is applied and what direction does the equilibrium shift? When adding NH3+ phenolphthalien+ NH4Cl What stress is applied and what direction does the equilibrium shift? When adding NH3+ phenolphthalien+ NaOH What stress is applied and what direction does the equilibrium shift? When adding NH3+ phenolphthalien+ HCl What stress is applied and what direction does the equilibrium shift?arrow_forward2. Consider the following equilibrium: H₂O(l) + HC₂H₂O₂(aq)=H₂O*(aq) + C₂H₂O₂(aq) Why will the addition of NaOH to a solution of acetic acid cause the concentration of the acetate ion to increase? ringarrow_forward
- 3 Complete the following condensation reactions by briefly explaining in each case the type of reaction taking place. iv. V. 1. NaOEt 2. H₂O/HCI 1. NaOEt 2. H₂O/HCIarrow_forwardIn reaction D: CuO + H2SO4= CUSO4 + H2O Students will occasionally use HNO3 (aq) instead of H2SO4 (aq) in reaction D, assuming that both strong acids will accomplish the same purpose. Briefly describe the results of this error.arrow_forwardIdentify the set of reaction conditions that best facilitates this reaction. Select the single best answer.arrow_forward
- (Use H3O+ instead of Ht.) 5. The value of Kb for methylamine, CH3NH2, is 4.20x104. Write the equation for the reaction that goes with this equilibrium constant.arrow_forwardI was super excited to get into the lab and make compound B from Compound A. I refluxed compound A in water and crossed my fingers. Son of a .... it turned out I made Compound C. What kind of reaction was this? Should I have used NaOH in water instead? $=$ H₂O Br OH B НО. 88arrow_forwardWhich of the following can inhibit nitrification? 1. Low HNO2 2. High NH3 3. High NH4+ 4. pH < 5 5. Low HCO3- 6. High HNO2 7. High NO2arrow_forward
- Explain why the pH of 0.1 M acetic acid (pH = 3) is higher than the pH of 0.1 M trifluoroacetic acid (pH = 1).arrow_forwardPart I. Write the hydrolytic reaction and equilibrium expression for the compounds below. 1. Propanoic acid (CH3CH2COOH) 2. Ethylamine (CH;CH2NH2)arrow_forwardCompute the amount of reagents which will be used for the preparation of the following reagents. Show your computation neatly. 500 mL of 0.1 M HCl Describe the preparation and storage of each reagents.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





