
An Introduction to Physical Science
14th Edition
ISBN: 9781305079137
Author: James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 4SA
An older type of thermostat used in furnace and heat pump control is shown in ● Fig. 5.21. The glass vial tilts back and forth so that electrical contacts are made via the mercury (an electrically
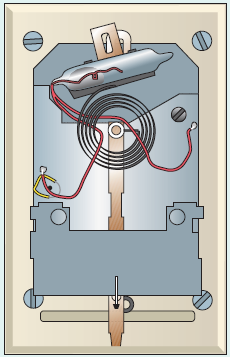
Figure 5.21 An Exposed View of a Thermostat See Short Answer Question 4.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
A 2-kW electric resistance heater submerged in 5-kg water is turned on and kept on for 10 min. During the
process, 300 kJ of heat is lost from the water. Solve for the temperature rise of water.
A hot water bottle is filled with either water or vegetable oil. It is placed in the microwave
until the temperature increases by 60°C.
Using the data below, answer the questions. Recall the specific heat equation:
energy = mass × SHC × temperature change
Mass of water in bottle
Mass of oil in bottle
.3 kg
.2 kg
Specific heat capacity of water ...4200 J/kg°C
Specific heat capacity of oil ....1600 J/kg°C
© Photos.com 2016
1. Calculate the energy stored in the water-filled bottle.
Answer:
An electric shower has a 1.5 kW heating element.
(a).How much heat energy can it give out in five minutes?
(b).If the element is used to heat 5 kg of water for 5 minutes, what would be the rise in temperature? (Specific heat capacity of water = 4180 J/kg).
Chapter 5 Solutions
An Introduction to Physical Science
Ch. 5.1 - We talk about temperature, but what does it...Ch. 5.1 - Are there any limits on the lowest and highest...Ch. 5.1 - Show that a temperature of 40 is the same on both...Ch. 5.2 - Prob. 1PQCh. 5.2 - Most substances contract with decreasing...Ch. 5.3 - What is specific about specific heat?Ch. 5.3 - Prob. 2PQCh. 5.3 - Prob. 5.2CECh. 5.3 - How much heat must be removed from 0.20 kg of...Ch. 5.4 - What are the three methods of heat transfer?
Ch. 5.4 - Prob. 2PQCh. 5.5 - Prob. 1PQCh. 5.5 - Prob. 2PQCh. 5.6 - In the ideal gas law, pressure is directly...Ch. 5.6 - Prob. 2PQCh. 5.6 - Prob. 5.4CECh. 5.7 - Prob. 1PQCh. 5.7 - Prob. 2PQCh. 5 - Prob. AMCh. 5 - Prob. BMCh. 5 - Prob. CMCh. 5 - Prob. DMCh. 5 - Prob. EMCh. 5 - Prob. FMCh. 5 - Prob. GMCh. 5 - Prob. HMCh. 5 - Prob. IMCh. 5 - Prob. JMCh. 5 - Prob. KMCh. 5 - Prob. LMCh. 5 - Prob. MMCh. 5 - Prob. NMCh. 5 - Prob. OMCh. 5 - Prob. PMCh. 5 - Prob. QMCh. 5 - Prob. RMCh. 5 - Prob. SMCh. 5 - Prob. TMCh. 5 - Prob. UMCh. 5 - Prob. VMCh. 5 - Prob. WMCh. 5 - Prob. XMCh. 5 - Prob. YMCh. 5 - Prob. 1MCCh. 5 - Which unit of the following is smaller? (5.2) (a)...Ch. 5 - Prob. 3MCCh. 5 - Prob. 4MCCh. 5 - Prob. 5MCCh. 5 - Prob. 6MCCh. 5 - Prob. 7MCCh. 5 - Which of the following has a definite volume but...Ch. 5 - If the average kinetic energy of the molecules in...Ch. 5 - When we use the ideal gas law, the temperature...Ch. 5 - Prob. 11MCCh. 5 - Prob. 12MCCh. 5 - When a bimetallic strip is heated, it bends away...Ch. 5 - Prob. 2FIBCh. 5 - Prob. 3FIBCh. 5 - Prob. 4FIBCh. 5 - Prob. 5FIBCh. 5 - Prob. 6FIBCh. 5 - Prob. 7FIBCh. 5 - The ___ phase of matter has no definite shape, and...Ch. 5 - Prob. 9FIBCh. 5 - In the ideal gas law, pressure is ___ proportional...Ch. 5 - Prob. 11FIBCh. 5 - Prob. 12FIBCh. 5 - When the temperature changes during the day, which...Ch. 5 - Prob. 2SACh. 5 - The two common liquids used in liquid-in-glass...Ch. 5 - An older type of thermostat used in furnace and...Ch. 5 - Heat may be thought of as the middleman of energy....Ch. 5 - When one drinking glass is stuck inside another,...Ch. 5 - Prob. 7SACh. 5 - What does the specific heat of a substance tell...Ch. 5 - When eating a piece of hot apple pie, you may find...Ch. 5 - Prob. 10SACh. 5 - When you exhale outdoors on a cold day, you can...Ch. 5 - Compare the SI units of specific heat and latent...Ch. 5 - Give two examples each of good thermal conductors...Ch. 5 - Prob. 14SACh. 5 - Prob. 15SACh. 5 - Thermal underwear is made to fit loosely. ( Fig....Ch. 5 - What determines the phase of a substance?Ch. 5 - Give descriptions of a solid, a liquid, and a gas...Ch. 5 - Prob. 19SACh. 5 - How does the kinetic theory describe a gas?Ch. 5 - Prob. 21SACh. 5 - Prob. 22SACh. 5 - Prob. 23SACh. 5 - In terms of kinetic theory, explain why a...Ch. 5 - Prob. 25SACh. 5 - Prob. 26SACh. 5 - Prob. 27SACh. 5 - Prob. 28SACh. 5 - What can be said about the total entropy of the...Ch. 5 - Prob. 30SACh. 5 - Prob. 31SACh. 5 - Prob. 1VCCh. 5 - Prob. 1AYKCh. 5 - Prob. 2AYKCh. 5 - Prob. 3AYKCh. 5 - Prob. 4AYKCh. 5 - Prob. 5AYKCh. 5 - Prob. 6AYKCh. 5 - When you freeze ice cubes in a tray, there is a...Ch. 5 - Prob. 8AYKCh. 5 - Prob. 1ECh. 5 - Prob. 2ECh. 5 - Prob. 3ECh. 5 - Prob. 4ECh. 5 - Researchers in the Antarctic measure the...Ch. 5 - Prob. 6ECh. 5 - A college student produces about 100 kcal of heat...Ch. 5 - Prob. 8ECh. 5 - A pound of body fat stores an amount of chemical...Ch. 5 - Prob. 10ECh. 5 - On a brisk walk, a person burns about 325 Cal/h....Ch. 5 - Prob. 12ECh. 5 - How much heat in kcal must be added to 0.50 kg of...Ch. 5 - Prob. 14ECh. 5 - (a) How much energy is necessary to heat 1.0 kg of...Ch. 5 - Equal amounts of heat are added to equal masses of...Ch. 5 - How much heat is necessary to change 500 g of ice...Ch. 5 - A quantity of steam (300 g) at 110C is condensed,...Ch. 5 - Prob. 19ECh. 5 - A fire breaks out and increases the Kelvin...Ch. 5 - A cylinder of gas is at room temperature (20C)....Ch. 5 - A cylinder of gas at room temperature has a...Ch. 5 - A quantity of gas in a piston cylinder has a...Ch. 5 - If the gas in Exercise 23 is initially at room...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- What is the role of “loose” electrons in heat conductors? (A) Loose electrons move quickly away from hot locations, making it impossible for energy to move and making the object a good insulator. (B) Loose electrons absorb energy, giving materials a high specific heat capacity. (C) Loose electrons vibrate and emit radiation that carries energy through the material at the speed of light. (D) Loose electrons transfer energy rapidly through a solid.arrow_forwardAir at 22°C is blown over a hot pipe with a surface area of 3.19 m2 to dissipate 927 W of heat energy. What is the minimum convection heat transfer coefficient that will ensure that the temperature of the pipe surface is less than 45°C [round your final answer to two decimal places]? T. Air, T.arrow_forwardQuestion 1 During a 30-minute period under clear skies, 2.0 x 10 J of total solar energy were incident upon Bunche Hall. Use this information on energy and time to complete the following tasks. Calculate the mean incident radiant flux over that 30-minute period. What are the units for the previous answer for Radiant Flux Kelvin Meters Microns J/s^2 J/s Watts W/m^2 Nanometers W/m Joules Fahrenheit Celsiusarrow_forward
- Questions: 2nd: Calculate the thermal resistance (R or R₂) of human skin with a thickness of 2 cm, per cm² of skin. ● thermal conductivity=0.627 N/(°C.s) ● • specific heat-c-3470 Nm/kg °C • density=1100 kg/m³ Rt - L KA where k is the materials conductivity [W.m³!.K-¹] L is the plane thickness [m] A is the plane area [m²]arrow_forwardSolve with the correct units 3. Determine the temperature for which a thermometer with degrees Fahrenheit is numerically twice the reading of the temperature in degrees Celsius.arrow_forwardQuestion 3.2+lab: Briefly describe the conditions when heat transfer in a heat exchanger reaches its maximum value.Explain the Fouling phenomenon. What is the effect of fouling on heat transfer rate?arrow_forward
- 3 JgpgenAf561PFw1CPObg/viewform?hr_submission=Chg106m4_QoSEA.. A 4 Which choice below that most simply describes the energy transformation that occurs when gasoline makes a car run? GASOLINE home er داری Electrical to Thermal Electrical to Radiant (Light) Chemical to Mechanical (Kinetic) Chemical to Kinetic (Mechanical) $ 4 F4 11 R F5 % 5 T F6 A 6 DELL F7 Y & 7 DL U 8 00 9 prt sc F10 O ) F11 I Parrow_forwardHow long does a 52 kW water heater take to raise the temperature of 170Lof water from 20°C to 37°C? The specific heat of water is 4186 J/kg-K. The density of water is 1.00 g/cm,arrow_forwardWhen you turn your hot water on in the shower, it takes a bit for the water to get warm. Some of this is because the water in the pipe is cold but some of it is because the water from the hot water heater must heat the copper plumbing pipe itself. Copper has a specific heat of 0.39R. How much energy does it take to heat 2 kilograms of copper pipe from 70°F to 130°F? Report your answer in Joules. (Hint: Note that you'll need to convert kJ to J and F to K.)arrow_forward
- A person consumes about 2600 Cal a day. 1 Cal = 4186 J, and 1 kWh = 860 Cal. What is this energy in kilowatt-hours? Express your answer using two significant figures.arrow_forwarda) A hot water heater in a residential home runs for an average of 3 hours per day with a heat energy input of 4.1 kW. What would be the annual cost for hot water in this home using an electric hot water heater if the cost of electricity is $0.13/kWh [round your final answer to zero decimal places]? b) A hot water heater in a residential home runs for an average of 3.2 hours per day with a heat energy input of 3.7 kW. What would be the annual cost for hot water in this home using a gas hot water heater if the cost of natural gas is $0.33/m3? The gas water heater can get 23 MJ of energy from 1 m3 of natural gas. [round your final answer to zero decimal places]?arrow_forwardYou have a Switch LED Lightbulb with the following information: It is equivalent to a 60 Watt incandescent lightbulb. It uses 12 Watts of electricity. It is estimated to last 25,000 hours. It is dimmable. It produces 800 lumens of light. It has a color temperature of 2,700K (warm white). Based on this information, calculate the lighting efficiency of this bulb and determine whether this is more or less efficient than a traditional incandescent lightbulb. Group of answer choices 0.015 lumens/Watt, more efficient 67 lumens/Watt, less efficient 0.015 lumens/Watt, less efficient 67 lumens/Watt, more efficientarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

University Physics (14th Edition)
Physics
ISBN:9780133969290
Author:Hugh D. Young, Roger A. Freedman
Publisher:PEARSON

Introduction To Quantum Mechanics
Physics
ISBN:9781107189638
Author:Griffiths, David J., Schroeter, Darrell F.
Publisher:Cambridge University Press

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Lecture- Tutorials for Introductory Astronomy
Physics
ISBN:9780321820464
Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina Brissenden
Publisher:Addison-Wesley

College Physics: A Strategic Approach (4th Editio...
Physics
ISBN:9780134609034
Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart Field
Publisher:PEARSON
Heat Transfer: Crash Course Engineering #14; Author: CrashCourse;https://www.youtube.com/watch?v=YK7G6l_K6sA;License: Standard YouTube License, CC-BY