
Concept explainers
(a)
Interpretation:
The name and identification of all the present groups or substituents should be determined in the following compound.
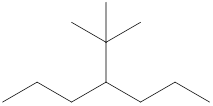
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms.
(b)
Interpretation:
The name and identification of all the present groups or substituents should be determined in the following compound.
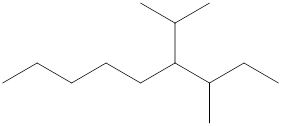
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that are double and triple bonds.
(c)
Interpretation:
The name and identification of all the present groups or substituents should be determined in the following compound.
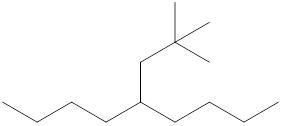
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that are double and triple bonds.
(d)
Interpretation:
The name and identification of all the present groups or substituents should be determined in the following compound.
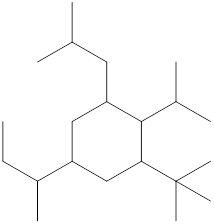
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that are double and triple bonds.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
- Give the complete IUPAC name and the common name for each of the following compounds:arrow_forwardThe following names are incorrect by IUPAC rules. Determine the correct IUPAC name for each compound. a. 2-Ethyl-2-pentene b. 4,5-Dimethyl-4-hexene c. 3,5-Cyclopentadiene d. l,2-Dimethyl-4-cyclohexenearrow_forwardThe correct IUPAC name for the following compound is: CH3CH2CCH2CH2CH2C1 CH2 1-Chloro-4-methylenehexane 1-Chloro-4-ethyl-4-pentene 5-Chloro-2-ethyl-1-hexene 5-Chloro-2-ethyl-1-pentene 2-(3-Chlorophyl)-1-butenearrow_forward
- Pick the best IUPAC name for the following molecule: Cl- OH O1-chloro-6-fluorocyclohexan-3-ol 1-chloro-2-fluorocyclohexanone O 3-chloro-4-fluorocyclohexan-1-ol O 1-chloro-2-fluorocyclohexan-5-olarrow_forwardWhat is the IUPAC name for the compound below? CH2CH3 CH;CHCHCH,CH3 ČH3 O 2-ethyl-3-methylpentane O 4-ethyl-3-methylpentane octane O 2,3-diethylbutane O 3,4-dimethylhexanearrow_forwardCyclopentane has a higher boiling point than pentane even though both compounds have the same number of carbons (49 °C vs. 36 °C). Can you suggest a reason for this phenomenon?arrow_forward
- Give the IUPAC name for each of the following: Part A CH3–CH2–CH,C–H Spell out the full name of the compound. Submit Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remainingarrow_forwardName the following organic compounds: compound CH3CH₂CH₂ 1 CH3CH₂CH₂-C- CH₂ CH₂ CH3 1 CH₂ - CH₂ name 4-ethyl-4-propylheptanearrow_forwardDraw the five isomers of C6H14 and write the IUPAC name of each compound.arrow_forward
- Name the following molecule. Remember to use dashes, commas and spaces as appropriate, to alphabetize your substituents, and to spell correctly! H3C CH2 CH H. H3C H2 H2 H2arrow_forwardDraw the 14 constitutional isomers of molecular formula C8H9Cl thatcontain a benzene ring.arrow_forwardWhat is the molecular formula of 5-chloro-1-ethylcyclohexene? O C8H12CI none of these O C8H13CI O C₂H₁6CI O C6H₁4 CIarrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





