
Interpretation:
The total number of neutrons present in the atom that has
Concept Introduction:
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
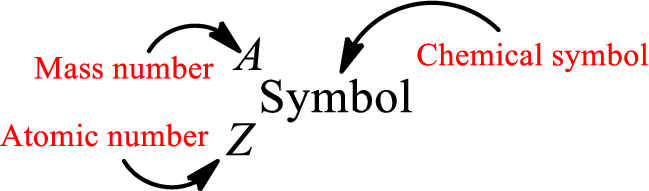
An element is a pure substance that cannot be broken by ordinary
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General, Organic, and Biological Chemistry
- Title:Complete the blank Description: Instructions: Fill in the blanks the number of atomic mass, atomic number, # of neutrons, and # of electrons of the given atom. 17. 9 Be 1. Mass number 4 2. Atomic number 3. # of neutrons 4. # of electrons Answer : 1. 2. 3. 4.arrow_forwardInstruction: Calculate the number of each particle in an atom that is missing in the table. SHOW +YOUR SOLUTION Name of Element Mass Atomic Protons Neutrons Electrons Element Symbol Number Number Boron В 11 5 6 Sodium 24 11 Y 89 39 Соpper 29 35arrow_forwardSelect the correct symbol for the atom or ion with 11 protons, 12 neutrons, and 11 electrons. a) 23Mg+ b) 23Na+ c)23Mg d)23Na e)22Na+ f)22Mgarrow_forward
- Ch.2 problems: 6. Tungsten is atomic number 74 on the Periodic Table. Supply the indicated information for a neutral isotope of tungsten-184, 184W. # of protons mass # # of electrons # of neutronsarrow_forwardhttps://course.apexlearning.com/public/activity/2 Options v No results arch L 2.2.3 Study: The Periodic Table Select each item in the left column and its match in the right column. Elements Number Fact The amount of elements proposed by Lavoisier in 1789 33 elements The number of different elements foundlin the human body 6 elements 99.9% of the matter in a living organism 63 elements 25 elements The number of known elements by the year 1869 Matches: 0 Tries: 0 RESET E PREVIOUS 2 of 23 NEXT > re to search DII ) F6 FS F7 Prt Scn F8 Home f9 End F10 PgUm % & ( ) 7 8 R Y | P ト %toarrow_forwardMATH PROBLEMS and SHORT ANSWER QUESTIONS - Show your Work: 10. For the Isotope 6731Ga A. What is the atomic number? B. What is the mass number? C. What is the number of protons? D. What is the number of neutrons? 1 Accessibility: Good to go a hparrow_forward
- Review I Constants I Periodic Ta Part A Two isotopes of gallium are naturally occurring, with Ga at 60.11% (68.93 amu) and Ga at 39.89% (70.92 amu). Calculate the atomic mass for gallium using the weighted 31 average mass method. Express your answer using four significant figures. amu Submit Request Answer Provide Feedback Next > - Previous Next MacBook Pro 80 $4 % & 5 6 7 8.arrow_forwardgen 13. Determine the number of neutrons for the following isotopes: A) Cr-53 Number of neutrons B) K-37 Number of neutronsarrow_forwardThe subatomic particles located in the nucleus with no overall charges are ________. a) protons b) electrons c) protons, neutrons, and electrons d)protons and neutrons e) neutronsarrow_forward
- Unit Test Review Active This is how manganese appears in the periodic table. 25 Mn Manganese 54.94 1 Mark this and return 2 0 0 0 0 4 Save and Exit 5 6 What is the arrow is pointing to? O O period symbol of manganese isotope symbol of manganese group symbol of manganese chemical symbol of manganese 7 8 19 Subnarrow_forwardReview I Constants I Periodic Table Complete the following table for two of the naturally occurring isotopes of potassium, which is an alkali metal: Atomic Symbol 39K 40K 41K 19- 19 Atomic Number Mass Number Number of Protons Number of Neutrons Number of Electrons Part E Write the number of electrons for K, K, 4K. 19 19 Enter your answers separated by commas. 39K, 40K, 4K: 19* ' 19*arrow_forwardA certain element consists of two stable isotopes. The first has a mass of 68.9 amu and a percent natural abundance of 60.4 %. The second has a mass of 70.9 amu and a percent natural abundance of 39.6 %. What is the atomic weight of the element? amu Submit Answer Try Another Version 3 item attempts remainingarrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
