
Concept explainers
Interpretation:
The number of different orbitals that contain electrons for the atom that has electronic configuration as
Concept Introduction:
Electronic configuration of an atom describes how many electrons are present in the shell. Many orbitals are present about the nucleus of an atom. In these orbitals the electrons do not occupy randomly. There are three rules for assigning the electrons to various shells, subshells, and orbitals. They are,
- The subshells are filled in increasing order of energy.
- In a subshell, the electrons occupy the orbital singly first in all orbitals before pairing up by the second electron. All the electrons that are in singly occupied orbitals have same spin.
- In a given orbital there cannot be more than two electrons and they have opposite spins.
Electronic configuration of an element is the one that gives information about how many electrons are present in each electron subshell of an atom. The electrons are added to the subshells in increasing order of energy. Electronic configurations are written in shorthand notation which uses a number‑letter combination. The shell is indicated by the number and subshell is indicated by the letter. Superscript that follows the subshell tells how many electrons are present in the subshell.
The order of filling up the electrons in the subshell is done as shown in the given figure below.
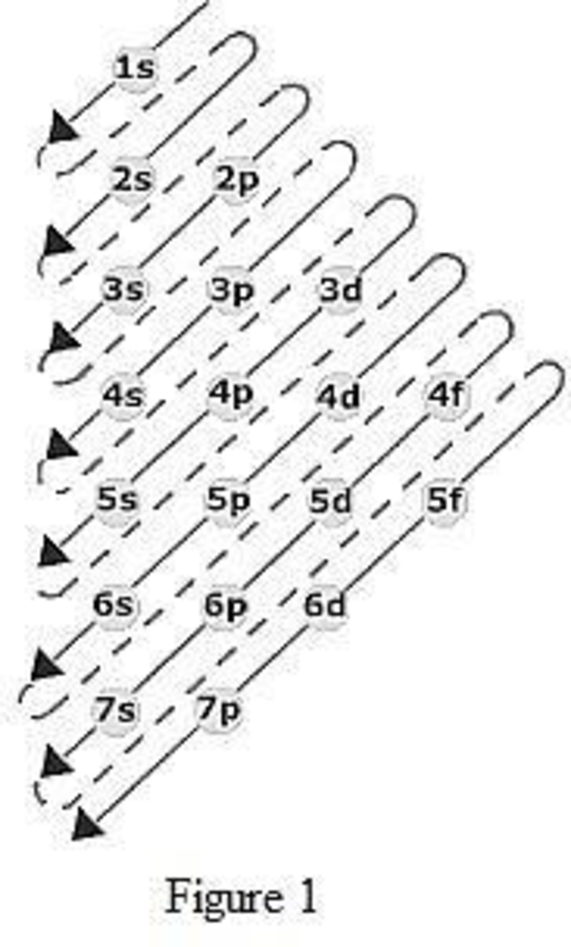
Orbital diagram is the one that gives information about the number of electrons present in the electron orbitals of an atom. The difference between electronic configuration and orbital diagram is that electronic configuration deals with the electrons occupancy in the subshell while orbital diagram deals with the electrons occupancy in the electron orbital. Electrons are paired up in the orbital only when all the orbitals in the subshell are singly filled.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General, Organic, and Biological Chemistry
- Infrared radiation falls in the wavelength region of 1.00×10-6 to 1.00×10-3 meters. What is the frequency of Infrared radiation that has a wavelength of 2.59x10-4 m. Frequency= Submit Answer Use the References to access important values if ne sec¹. 4 question attempts remaining 4arrow_forward[Refe Which of the following is the ground-state electron configuration of a C ion? O 1s 2s O 1s 2s 2p O 1s O 1s 25 O 1s 2s 2p° O 1s 2s 2p Submit Answer Try Another Version 6 item attempts remainingarrow_forward6.7.2 The energy of an orbital in the hydrogen atom depends on_______________.a) n, l, and ml c) n only e) ml onlyb) n and l d) l onlyarrow_forward
- ▼ Part A Which electron transition produces light of the highest frequency in the hydrogen atom? 4p → 1s О 3р - 1s 2p 1s 5p →→ 1s Submit Provide Feedback Request Answerarrow_forwardHow many states correspond to the n = 6, l = 5 level of hydrogen? a) 11 b) 12 c) 22 d) 24 e) 33 f) None of the other responses are correctarrow_forwardPart A The Li nucleus has an excited state 0 48 MeV above the ground state. What wavelength gamma photon is emitted when the nucleus decays from the excited state to the ground state? m Submit Request Answer Provide Feedbackarrow_forward
- 三 Objective Knowledge Check Question 12 Rank the effective nuclear charge Z experienced by a valence electron in each of these atoms: z* experienced by a valence electron. atom An atom of magnesium. [(pick one) An atom of sodium. (pick one) An atom of silicon. (pick one) v An atom of aluminum. (pick one) I Don't Know Submit O 2021 McGraw Hill LLC. ♡ Type here to searcharrow_forwardDI Chapter 2 Multiple Choice Question 69 Part A What is the frequency of light with a wavelength of 0.440 um? O 2.55 x 1013 sec1 O 5.93 x 1011 sec1 O 6.81 x 1014 sec O 7.28 x 1014 sec1 O 6.81 x 1011 sec1 Submit Request Answer Provide Feedback P Type here to search ? f2 f3 f4 esc 15arrow_forward四 %23 Tue Oct 26 8 X P Pearson Sign In In Hwk#6 (page 7 of 16) me + x p?attempt%3D1047279&cmid=786108&page3D6 Irini Konstantinou emistry I F21) Search courses Quick Links 9# A certain metal has a work function of 315.0 kJ per mole of e-. What wavelength of electromagnetic radiation (in nm) must be directed at the surface in order for electrons to be ejected with a de Broglie wavelength of 13.83 angstroms (A) 1 Á = 1×100 metersarrow_forward
- PLS HELP VERY NECESSARY: Q2) Which subshell contains only one orbital? a) 4d b) 6f c)3d d)2s e)1p no explanation required, only answer thanks!arrow_forward6.5.1 What is the minimum uncertainty in the position of anelectron moving at a speed of 4 × 106 m/s ± 1 percent?(The mass of an electron is 9.11 × 10−31 kg.)a) 2 × 10−8m d) 7 × 10−8m b) 1 × 10−9m e) 1 × 10−12 mc) 6 × 10−9marrow_forward20 Complete the electron configuration for Br. Macmillan Learning electron configuration: [Ar] 1s² 2s² 2p 3s² 3p6 4s² 3d¹04p5 Incorrect Attempt 4arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
