
Concept explainers
a. Consider the malate dehydrogenase reaction, which is part of the citric acid cycle:
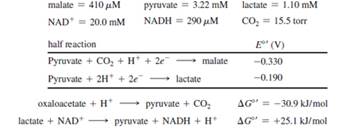
malate + NAD+
In yeast mitochondria, where the pH= 8.1, this reaction is exergonic only at low oxaloacetate concentrations. Assuming a pH = 8.1, a temperature of 37o C, and the steady-state concentrations given below, calculate the maximum concentration of oxaloacetate at which the reaction will still be exergonic.
b. How would a drop in pH affect
c. How would an increase in intracellular malate levels affect
d. How would an increase in intracellular pyruvate levels affect
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Biochemistry: Concepts and Connections (2nd Edition)
- a. Some of the acetyl-CoA used in the citric acid cycle is produced from pyruvate. List the reactants and products of this reaction. The reactants are pyruvate and The products are acetyl-CoA and In the process, a(n) is to b. Name an enzyme complex that catalyzes this reaction and list its negative effectors. is the enzyme complex that catalyzes this reaction. Its negative effectors are andarrow_forward. Because of the position of arsenic in the periodic table, arsenate (AsO}-) is chemically similar to inorganic phosphate and is used by phosphate-requiring enzymes as an alternative substrate. However, organic arsenates are quite unstable and spontaneously hydrolyze. Arsenate is known to inhibit ATP production in glycolysis. Identify the target enzyme, and explain the mechanism of inhibition.arrow_forwardA. Explain why it is important that an energy producing pathway contains at least one regulatory enzyme, you can use either glycolysis or TCA enzymes to discuss this answer B. The first step in glycolysis involves the conversion of glucose to glucose-6- phosphate. Briefly explain how this reaction occurs as it is an endergonic reaction. C. Why is it important that the cell has a number of different high-energy biomolecules?arrow_forward
- Consider oleic acid (18:1∆9): 1. How many NADH will be produced from complete oxidation of this fatty acid? 2. How many FADH2 will be produced from complete oxidation of this fatty acid? 3. Total number of ATP produced from NADH in complete oxidation of oleic acid (exclude transport cost of the fatty acid)? 4. Total number of ATP produced from FADH2 in complete oxidation of oleic acid (exclude transport costs of the fatty acid)? Please provide how you got them.arrow_forwardA. The inhibitor constants for three inhibitors of por- cine citrate synthase are summarized in the table on the right. The compounds were all determined to bind in the active site as competitive inhibitors of acetyl-CoA. Because they bind as competitive inhibitors, all three inhibitors must exhibit structural similarity to some part of acetyl-CoA. Look up in the textbook the structural formu- las for Coenzyme A, ATP, and NADH. What is the largest structural fragment of each inhibitor that is responsible for competitive inhibition? Draw the molecular fragment common to each inhibitor that competes with the binding of acetyl-CoA in the active site of citrate synthase. Bromoacetyl-CoA ATP NADH K₁ (μM) 25.7 6800 8300 B While the inhibitor constants listed in part (b) above were determined in vitro for purified citrate synthase, does their inhibitory action have any relevance to the flux of metabolites through the TCA cycle in vivo? If so, explain.arrow_forwardA. Identify different types of organic reaction mechanims in the followingmetabolic pathways.1. Catabolism of triacylglycerols- beta-oxidation pathway2. Biosynthesis of fatty acids from Acetyl CoA3. Glycolysis (from glucose to two molecules of pyruvate)4. Conversion of Pyruvate to Acetyl CoA5.Citric acid cycle6. Gluconeogensis pathway (pyruvate to glucose) B. Identify at most 5 organic reactions for each metabolic pathway.arrow_forward
- (a) Consider the oxidation of malate to oxaloacetate by NAD*: malate + NAD+ → oxaloacetate + NADH + H+ In yeast mitochondria, where the pH = 8.1, this reaction is exergonic only at low oxaloacetate concentrations. Assuming a pH = 8.1, a temperature of 37 °C, and the steady-state concentrations given below, calculate the maximum concentration of oxaloacetate at which the reaction will still be exergonic. malate + NAD*→ oxaloacetate + NADH + H* lactate + NAD →→ pyruvate + NADH + H+ half reaction Pyruvate + 2H+ + 2e → lactate Pyruvate + CO₂ + H + 2e → malate Intracellular steady state concentrations: malate = 410 μM; NAD = 20.0 mM; pyruvate = 3.22 mM; NADH = 290 μM; AG=+29.7 kJ/mol AG¹ = +25.1 kJ/mol E° (V) - 0.190 - 0.330 lactate 1.1 mM CO₂ = 15.5 torrarrow_forwardGiven the following information, calculate the physiological ΔG of the isocitrate dehydrogenase reaction at 25°C and pH 7.0: [NAD+]/[NADH] = 8, [α-ketoglutarate] = 0.1 mM, and [isocitrate] = 0.02 mM. Assume standard conditions for CO2 (ΔG°′ is given in Table). Is this reaction a likely site for metabolic control?arrow_forwardDescribe the mechanism of a-chymotrypsin. Explain the roles of constituents of the catalytic triad, their modes of catalysis, and the significance of the oxyanion hole in the catalysis.arrow_forward
- Page of 6 ZOOM + name: 3. In the last reaction of the citric acid cycle, malate is dehydrogenated to regenerate the oxaloacetate necessary for the entry of acetyl-CoA into the cycle: L-Malate + NAD+ → oxaloacetate + NADH + H* AG'° = 30.0 kJ/mol (a) Calculate the equilibrium constant for this reaction at 25 °C. (b) Because AG°' assumes a standard pH of 7, the equilibrium constant calculated in (a) corresponds to [oxaloacetate][NADH] Keq [L-malate][NAD*] The measured concentration of L-malate in rat liver mitochondria is about 0.20 mM when [NAD*]/[NADH] is 10. Calculate the concentration of oxaloacetate at pH 7 in these mitochondria. (c) To appreciate the magnitude of the mitochondrial oxaloacetate concentration, calculate the number of oxaloacetate molecules in a single rat liver mitochondrion. Assume the mitochondrion is a sphere of diameter 2.0 microns.arrow_forwardConsider the complete oxidation of one mole of simple TAG containing behenic acid residues (22:0). II. What is the net ATP yield for the complete oxidation of all the fatty acid residues of the simple TAG? (Note: glycerol backbone is not included)arrow_forwardA. Identify different types of organic reaction mechanims in the following metabolic pathways.4. Conversion of Pyruvate to Acetyl CoA5.Citric acid cycle6. Gluconeogensis pathway (pyruvate to glucose) B. Identify at most 5 organic reactions for each metabolic pathway.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





