
Concept explainers
(a)
Interpretation: The explanation of the shape of the given graph is to be stated.
Concept introduction:
(a)
Answer to Problem 104AE
The graph show increasing pattern as mass of
Explanation of Solution
The given graph is as follows.
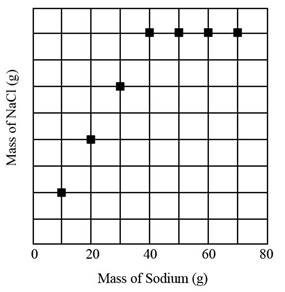
Figure 1
The graph constitutes
The graph shows an increasing pattern at first as the mass of
Therefore, the graph show increasing pattern till fourth sample as mass of
(b)
Interpretation: The mass of
Concept introduction: Stoichiometric coefficients proportional to the respective chemical species can be correlated so as to compute their moles. The corresponding mass of these chemical species can then be worked out via their molar masses. The former concept is designated as stoichiometry. This concept can also be utilized to acquire limiting as well as excess reactants in given reaction.
(b)
Answer to Problem 104AE
The mass of
Explanation of Solution
The balanced reaction of
The given mass of
The number of moles of
The molar mass of
Substitute the values in equation (1).
The above reaction shows that
The mass of
The molar mass of
Substitute the values in equation (2).
Therefore, the mass of
(c)
Interpretation: The mass of
Concept introduction: Stoichiometric coefficients proportional to the respective chemical species can be correlated so as to compute their moles. The corresponding mass of these chemical species can then be worked out via their molar masses. The former concept is designated as stoichiometry. This concept can also be utilized to acquire limiting as well as excess reactants in given reaction.
(c)
Answer to Problem 104AE
The mass of
Explanation of Solution
The balanced reaction of
The above reaction shows that
The mass of
The graph shows that after fourth addition that is when
The mass of
Therefore, the mass of
(d)
Interpretation: The mass of
Concept introduction: Stoichiometric coefficients proportional to the respective chemical species can be correlated so as to compute their moles. The corresponding mass of these chemical species can then be worked out via their molar masses. The former concept is designated as stoichiometry. This concept can also be utilized to acquire limiting as well as excess reactants in given reaction.
(d)
Answer to Problem 104AE
The mass of
Explanation of Solution
The balanced reaction of
The given mass of
The molar mass of
Since, the maximum amount of
The number of moles of
The above reaction shows that
The molar mass of
Substitute the values in equation (2) to calculate the mass of
Therefore, the mass of
(e)
Interpretation: The left over reactants and their masses in parts b and d are to be calculated.
Concept introduction: Stoichiometric coefficients proportional to the respective chemical species can be correlated so as to compute their moles. The corresponding mass of these chemical species can then be worked out via their molar masses. The former concept is designated as stoichiometry. This concept can also be utilized to acquire limiting as well as excess reactants in given reaction.
(e)
Answer to Problem 104AE
The leftover reagent in part b is
Explanation of Solution
The balanced reaction of
The above reaction shows that
The mass of
The graph shows that
The given mass of
The mass of
The mass of
Substitute the values in equation (3).
The graph shows that in part d,
The mass of
Substitute the values in equation (4).
Therefore, the leftover reagent in part b is
Want to see more full solutions like this?
Chapter 3 Solutions
Chemical Principles
- When calcium carbonate is heated strongly, it evolves carbon dioxide gas. CaCO3(s)CaO(s)+CO2(g) 25 g of CaCO3 is heated, what mass of CO2would be produced? What volume would this quantity of CO2 (CU at STP?arrow_forwardA power plant is driven by the combustion of a complex fossil fuel having the formula C11H7S. Assume the air supply is composed of only N2 and O2 with a molar ratio of 3.76:1.00, and the N2 remains unreacted. In addition to the water produced, the fuels C is completely combusted to CO2 and its sulfur content is converted to SO2. In order to evaluate gases emitted at the exhaust stacks for environmental regulation purposes, the nitrogen supplied with the air must also be included in the balanced reactions. a Including the N2 supplied m the air, write a balanced combustion equation for the complex fuel assuming 100% stoichiometric combustion (i.e., when there is no excess oxygen in the products and the only C-containing product is CO2). Except in the case of N2, use only integer coefficients. b Including N2 supplied in the air, write a balanced combustion equation for the complex fuel assuming 120% stoichiometric combustion (i.e., when excess oxygen is present in the products and the only C-containing product is CO2). Except in the case of use only integer coefficients c Calculate the minimum mass (in kg) of air required to completely combust 1700 kg of C11H7S. d Calculate the air/fuel mass ratio, assuming 100% stoichiometric combustion. e Calculate the air/fuel mass ratio, assuming 120% stoichiometric combustion.arrow_forwardMagnesium will react with steam, forming magnesium hydroxide and hydrogen gas, as shown in the reaction below. Mg(s) + 2 H2O(g) → Mg(OH)2(s) + H2(g) Consider an experiment in which 81.7 g of steam and 35.2 g of magnesium are allowed to react in a sealed reaction vessel. What mass of Mg(OH)2 will be created?arrow_forward
- What volume of hydrogen sulfide is required to produce 162 liters of sulfur dioxide according to the following reaction? (All gases are at the same temperature and pressure.) hydrogen sulfide (g) + oxygen(g) → water (1) + sulfur dioxide (g) Volume= Larrow_forward1. A sample of tin(II) nitrate, Sn(NO3)₂ (molar mass=242.71 g/mol), is heated in an evacuated cylinder (i.e. P=0 atm) with a volume of 6.72 L. The salt completely decomposes when heated, according to the equation: 2Sn(NO3)₂ (s) → 2SnO(s) + 4NO₂(g) + O₂(g) If the pressure in the cylinder after decomposition and cooling to a temperature of 277 K is 0.326 atm, what was the mass of the sample? Assume that the volume of solid chemicals involved in the reaction have negligible volume. A) 46.8 g B) 9.37 g C) 4.68 g D) 23.4 g E) 46.8 garrow_forwardNitroglycerin, a very unstable compound (molecular structure shown below), is a liquid at room temperature. When heated to 218.0°C or subjected to shock, it rapidly decomposes into four different gases. Liquid nitroglycerin has a density of approximately 1.60 g/mL. The nitroglycerin decomposition reaction equation is: 4 C3H5N3O9 (1)→ 12 CO₂(g) + 10 H₂O(g) + 6 N₂(g) + O₂(g) Assume that when heated to 218.0°C, 1.00 lb of nitroglycerin explodes heating the product gases to 750.0°C. Determine the total gas volume, in L, produced by the explosion. Assume that the pressure generated by the explosion is 7.50 atm. -O H₂C- H₂C -O -CH O N nitroglycerinarrow_forward
- 15. What volume (in L) of cobalt can be produced when 55.0 kg of colbalt(III) oxide reacts with 5.00 L of carbon? How many liters of excess reactant is leftover? (dCo(s) = 8.90 g cm; dcs) = 2.26 g cm -) Co,O3 (s) + C(s) → Co(s) + CO;(g) A: 4.4; 2.36arrow_forwardOne method used in the production of H2 gas is to put the coal into an incomplete combustion reaction with air and water vapor in the combustion chamber (water gas production). A simplified reaction can be written as:2C + 1 H2O + a (O2+3.76N2) = 1C0 + 1CO2 + 1H2 + 3.76·a·N2 Here, 02+3.76N2 represents the amount of air and is multiplied by the coefficient "a".a) The value of the coefficient "a" for this reaction b) The amount of air to be used for 1 kg of pure carbon (C) (total amount of 02 and N2) (kg-air/kg-carbon) Hint: Consider how many kilograms 1 kmol "C" weighs.arrow_forwardIf 14.0 LL of hydrogen chloride gas reacts with 49.5 LL of oxygen gas, what is the volume of chlorine gas produced? Assume all gases are at the same temperature and pressure. HCl(g)+O2(g)→Cl2(g)+H2O(g)arrow_forward
- The design of air-bag depends on stoichiometric precision. The following reaction takes place when the air-bag is inflated. 2 NaN3 (s) → 2 Na(s) + 3 N2 (g) 6Na(s) + Fe2O3(s) → 3NA2O(s) + 2Fe Assume that 43.4 L of nitrogen gas are needed to inflate an air bag to the proper size. (A) How many grams of NaN3 must be included in the air-bag to generate this amount of nitrogen gas? (The density of nitrogen gas at this temperature is 0.916 g/L). (B) How much Fe2O3 (in grams) must be added to the air-bag for this amount of NaN3? Use the following molar masses: N2 = 28.02 g/mol NaN3 = 65.01 g/mol Fe203 = 159.69 g/mol Type only the numerical answers in the answer box grams of NaN3 grams of Fe2O3 =arrow_forwardThe human body burns glucose (C6H₁2O6) for energy according to this chemical reaction: C6H12O6 +60₂-6CO₂ + 6H₂O The products of the reaction are carbon dioxide (CO₂) and water (H₂O). Interestingly, all of the carbon dioxide and much of the water exits the body through the lungs: on every breath, the average person exhales 500. mL of air, which is typically enriched to 4% CO₂ and 5% water vapor by volume. In short, when a person loses weight by dieting, the weight that is lost actually departs his body as a gas, every time he exhales. Each kilogram of body fat lost requires exhaling about 2.9 kg of carbon dioxide. Calculate how many breaths it takes an average person to "exhale" 1.00 kg of fat. Round your answer to the nearest thousand. You'll need to know that the density of CO₂ is 2.0 kg/m³. 0 ☐ ☐x10 Xarrow_forwardThe density of F2 gas is 1.70 g/L.How many moles of F2 gas are present in a 475 L sample of a gas mixture that is 4.00 % fluorine and 96.00 % helium?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


