
Concept explainers
(a)
Interpretation: To classify carbamoyl phosphate in terms of nitrogen content as
Concept introduction: Carbamoyl phosphate is an intermediate or one of the fuel for the urea cycle. Ammonium ion produced via oxidative deamination reaction is converted into the carbamoyl phosphate which then enters the urea cycle.
The carbamoyl phosphate formation reaction is carried out in the mitochondrial matrix. Carbamoyl is the prefix used to represent an amide group. Thus carbamoyl phosphate is a molecule that contains a phosphate group attached to an amide
Depending upon the number of nitrogen atom present in the structure of the compound it can be classified as
(a)
Answer to Problem 26.75EP
Carbamoyl phosphate is a
Explanation of Solution
Carbamoyl phosphate and aspartate are fuel for the urea cycle. The structure of carbamoyl phosphate is:
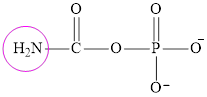
It contains only one nitrogen atom thus carbamoyl phosphate is characterized as
(b)
Interpretation: To classify glutamate in terms of nitrogen content as
Concept introduction: Glutamate is conjugate anion of glutamic acid. Glutamic acid is an amino acid. Depending upon the number of nitrogen atom present in the structure of the compound it can be classified as
(b)
Answer to Problem 26.75EP
Glutamate is a
Explanation of Solution
The structure of glutamate is:
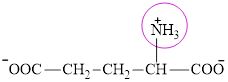
It contains only one nitrogen atom thus glutamate is characterized a
(c)
Interpretation: To characterize urea in terms of nitrogen content as
Concept introduction: The nitrogenous product of protein
Depending upon the number of nitrogen atom present in the structure of the compound it can be classified as
(c)
Answer to Problem 26.75EP
Urea is a
Explanation of Solution
The structure of urea is:
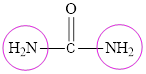
The structure of urea suggests the presence of two
(d)
Interpretation: To characterize citrulline in terms of nitrogen content as
Concept introduction: Citrulline is a nonstandard amino acid and is an inetrmediate in the the urea cycle. It is encountered at step 2 of the urea cycle and undergo condensation reaction with aspartate to give argininosuccinate.
Depending upon the number of nitrogen atom present in the structure of the compound it can be classified as
(d)
Answer to Problem 26.75EP
Citrulline is a
Explanation of Solution
The structure of citrulline is:
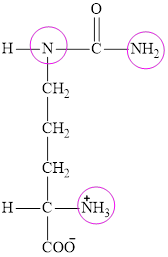
Citrulline contains three nitrogen atoms and thus it is characterized as a
Want to see more full solutions like this?
Chapter 26 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Which of the following is a degradation product of both cytidine and deoxycytidine? a. β-alanine b. β-aminoisobutyrate c. succinyl-CoA d. uric acidarrow_forwardThe reaction of glutamate and NH4* to yield glutamine is catalyzed by. a) Uridylyltransferase b) Adenylyltransferase c) Glutamate synthase d) Glutamine synthasearrow_forwardDefine the following terms: a. ketogenic amino acid b. glucogenic amino acid c. L-amino acid oxidase d. Krebs urea cycle e. CPSIarrow_forward
- The immediate donors of the nitrogen atoms of urea are: a. Aspartate and glutamate b. Glutamate and carbamoyl phosphate c. Aspartate and carbamoyl phosphate d. Glutamine and aspartatearrow_forwardWhich of the following condenses acyl and malonyl groups? a) Acetyl co-A ACP transacetylase b) Malonyl co-A ACP transferase c) ß-ketoacyl ACP synthase d) Acyl carrier proteinarrow_forwardAlkaptonuria is a rare organic aciduria involving a deficiency in homogentistic acid oxidase resulting in accumulation of homogentisic acid which is an intermediate in the degradative pathway of which of the following amino acids? a. Threonine b. Alanine c. Tyrosine d. Valinearrow_forward
- Which of the following amino acids can act as nucleophiles in a protease active site (choose all that apply): a) Cysteine b) Alanine c) Leucine d) Serine e) Threoninearrow_forwardWhy must nitrogen fixation occur in an anoxic (i.e., oxygen-free) environment? a)Because H2 is an additional product of nitrogen fixation, and the combination of H2 and O2 would cause the cell to combust. b)Oxygen poisons the dinitrogenase enzyme by entering the FeMo cluster c)Oxygen competes as an electron acceptor in the ETC, so electrons cannot be passed to N2. d)Oxygen "steals" electrons from the dinitrogenase reductase, producing free radicals.arrow_forwardDefine the following terms: a. oxidoreductase b. lyase c. ligase d. transferase e. isomerasearrow_forward
- Cyanide makes a bond to cytochrome in the electron transport chain located in the mitochondria, rendering the electron transport chain useless. The cyanide ingestion in death due to which of the following? a) Starvation, due to an inability to create glucose.b) Build up of lactic acid.c) Inability to take in oxygen through the lungs.d) Cellular death from lack of energy.arrow_forwardWhich enzyme is responsible for the cyclization of a glutamate derivative forming a proline precursor? A、AIR synthetase B、semialdehyde cyclase C、No enzyme is required D、IMP synthasearrow_forwardWhich of following is purely Ketogenic amino acid? a. Tyrosine b. Leucine c. Phenylalanine d. Tryptophanarrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





