
Concept explainers
(a)
Interpretation: To determine whether glutamate can be converted to
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
During transamination reaction the new keto acid formed has carbon skeleton similar to the carbon skeleton of the reacting amino acid and the new amino acid formed has the carbon skeleton similar to the carbon skeleton of the reacting keto acid.
(a)
Answer to Problem 26.32EP
Yes, glutamate can be converted to
Explanation of Solution
Glutamate is an amino acid and its structure is:
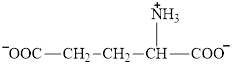
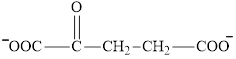
Both glutamate and
(b)
Interpretation: To determine whether
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
During transamination reaction the new keto acid formed has carbon skeleton similar to the carbon skeleton of the reacting amino acid and the new amino acid formed has the carbon skeleton similar to the carbon skeleton of the reacting keto acid.
(b)
Answer to Problem 26.32EP
Yes,
Explanation of Solution
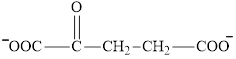
Glutamate is an amino acid and its structure is:
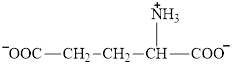
Both glutamate and
(c)
Interpretation: To determine whether glutamate can be converted to aspartate via a transamination reaction or not.
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
During transamination reaction the new keto acid formed has carbon skeleton similar to the carbon skeleton of the reacting amino acid and the new amino acid formed has the carbon skeleton similar to the carbon skeleton of the reacting keto acid.
(c)
Answer to Problem 26.32EP
No, glutamate cannot be converted to aspartate by transamination.
Explanation of Solution
Glutamate is an amino acid and its structure is:
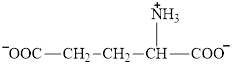
Aspartate is an amino acid and its structure is:
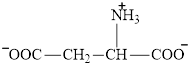
Transamination reaction involves the exchange of an amino group from an
(d)
Interpretation: To determine whether
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
During transamination reaction the new keto acid formed has carbon skeleton similar to the carbon skeleton of the reacting amino acid and the new amino acid formed has the carbon skeleton similar to the carbon skeleton of the reacting keto acid.
(d)
Answer to Problem 26.32EP
No,
Explanation of Solution
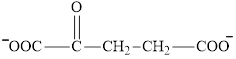
Oxaloacetate is a keto acid and its structure is:
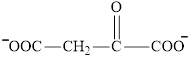
Transamination reaction involves the exchange of an amino group from an
Want to see more full solutions like this?
Chapter 26 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- The conversion of glutamate to alpha-ketoglutarate is an example of oxidative deamination. Give the complete chemical equation of the said pathway. What happens to the second product of this reaction?arrow_forwardExplain the chemical change that occurs in converting kynurenine (a product of tryptophan degradation) to kynurenate, a reaction in which α-ketoglutarate is transformed to glutamate.arrow_forwardPhosphoglycerate mutase (PGM) catalyzes the interconversion of 3-phosphoglycerate (3PG) and 2-phosphoglycerate (2PG) in the glycolytic and gluconeogenic pathways. a) To what enzyme class does PGM belong? b) There are two distinct classes of PGM, one which is dependent on 2,3-bisphosphoglycerate (2,3-BPG), dPGM, and one which is not, iPGM. dPGM uses acid base chemistry and a phosphorylated histidine residue to interconvert 3PG and 2PG. The dPGM reaction proceeds with formation of 2,3-BPG as an intermediate. Propose a mechanism for the dPGM-catalyzed conversion of 3PG to 2PG that is consistent with this information. c) What is the purpose of 2,3-BPG (i.e., why does dPGM require it)?arrow_forward
- Fatty acids are converted to their coenzyme A esters in a reversible reaction catalyzed by acyl-CoA synthetase: R-COO +ATP +COA R-C-COA +AMP + PPi a) The reaction involves two steps the first of which forms an enzyme-bound intermediate identified as the mixed anhydride of the fatty acid and AMP: R-C-O-P-O-nibose-adenine Write two chemical equations coresponding to the two steps of the reaction catalyzed by the synthetase. b) The acyl-CoA synthetase reaction as written above is readily reversible. How might the reaction be made to favor formation of fatty acyl-CoA? Write within the box. Anything outside the box will not be graded. From thearrow_forwardUDP-glucuronosyltransferase enzymes bind the organic compound UDP-glucuronic acid (UDP-GA) in order to catalyse the transfer of a glucuronic acid group from UDP-GA to a drug molecule, releasing UDP from the active site as a product. UDP is then regenerated by the activity of another enzyme. What terms could be used to describe UDP-GA?arrow_forwardPyridoxal phosphate acts as an intermediate carrier of amino groups during transamination reactions. Write a series of reactions to show the role of pyridoxal phosphate in the transamination reaction involving alanine and α-ketoglutarate.arrow_forward
- Assume that the 2nd carbon atom of pyruvate is labeled with C14 and then the labeledpyruvate is allowed to undergo metabolism in liver cells.a) Write down the metabolic reactions that allow the C14 carbon atom to be incorporatedinto glutamine.b) Please indicate which carbon atom in glutamine is labeled as C14.arrow_forwardWhich of the following statements about the transamination and deamination steps of amino acid degradation is true? (A) a-ketoglutarate is always formed during a transamination between an amino acid and glutamate. (B) Transamination reactions produce glutamate that is deaminated after entering the urea cycle. (C) Free ammonia is removed from glutamate using glutamate dehydrogenase and NAD+ as an oxidizing agent. (D) The free NH4+ that is removed from glutamate during the deamination reaction is used to form glucose.(E) The carbon backbone that results from transamination enters the mitochondria to be used in the urea cycle.arrow_forwardThe PDH complex is a logical point of regulation in metabolism, as it links two major catabolic processes. Answer the following regarding the complex: a) Explain the advantage of E1, E2 and E3 working as a complex as opposed to separately. b) Explain the purpose of each of the three enzymes and their associated cofactors. c) NADH can inhibit the PDH complex directly or indirectly. What is the purpose of inhibition by NADH? d) Explain the differences between direct and indirect inhibition by NADH. Be sure to indicate the components involved and the mechanism of inhibition (impact on target component).arrow_forward
- Why would glutamate utilize the oxidative deamination pathway over transamination – especially if removal of NH4+ requires ATP? Would it not be favorable for the body to just use transamination?arrow_forwardWhat is alpha keto glutarate dehydrogenase complex?. explain very briefly.arrow_forwardConsidering that triacylglycerols get hydrolyzed in the small intestine because of pancreatic lipases, write the reactions using structural formulas by which a 7-carbon fatty acid is activated and oxidized in the liver of this patient. Indicate cofactors and underscore the final CoA products.arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





