
Concept explainers
(a)
Interpretation: To determine whether glutamate and aspartate could function as the two reactants in a transamination reaction or not.
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
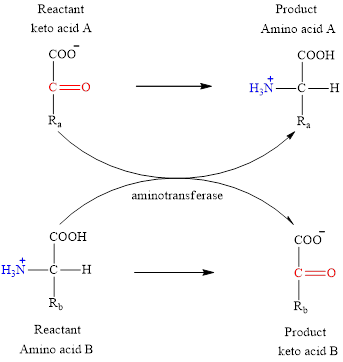
The general structure of an amino acid is:
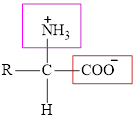
Here,
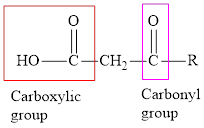
An acid containing both carbonyl and carboxyl
(a)
Answer to Problem 26.34EP
No, glutamate and aspartate cannot function as the reactants in a transamination reaction.
Explanation of Solution
Glutamate is an amino acid and its structure is:
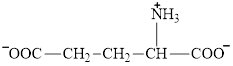
Aspartate is an amino acid and its structure is:
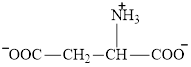
The two reactants in transamination reaction are a keto acid and an amino acid. Both glutamate and aspartate are amino acids thus they cannot function as reactants in a transamination reaction. For a transamination reaction to take place there must be one amino acid present along with a keto acid.
(b)
Interpretation: To determine whether aspartate and
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an

The general structure of an amino acid is:

Here,
An acid containing both carbonyl and carboxyl functional group is known as a keto acid. A general representation of a keto acid is:

(b)
Answer to Problem 26.34EP
Yes, aspartate and
Explanation of Solution
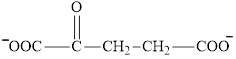
Aspartate is an amino acid and its structure is:
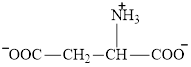
Transamination reaction involves the exchange of an amino group from an
(c)
Interpretation: To determine whether succinate and
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an

The general structure of an amino acid is:

Here,
An acid containing both carbonyl and carboxyl functional group is known as a keto acid. A general representation of a keto acid is:

(c)
Answer to Problem 26.34EP
No, succinate and
Explanation of Solution
Succinate is a diacid acid and its structure is:
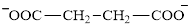
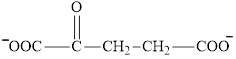
The two reactants in transamination reaction are a keto acid and an amino acid.
(d)
Interpretation: To determine whether glutarate and aspartate could function as the two reactants in a transamination reaction or not.
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
The general reaction to illustrate transamination is as follows:

The general structure of an amino acid is:

Here,
An acid containing both carbonyl and carboxyl functional group is known as a keto acid. A general representation of a keto acid is:

(d)
Answer to Problem 26.34EP
No, glutarate and aspartate cannot function as the reactants in a transamination reaction.
Explanation of Solution
Aspartate is an amino acid and its structure is:
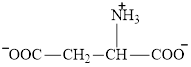
Glutarate is a diacid and its structure is:
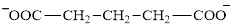
The two reactants in transamination reaction are a keto acid and an amino acid. Aspartate is an amino acid but glutarate is not a keto acid. For a transamination reaction to take place there must be one keto acid present along with an amino acid. Thus, glutarate and aspartate cannot function as the reactants in a transamination reaction.
Want to see more full solutions like this?
Chapter 26 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Transamination reactions have been described as ping-pong reactions. Use the reaction of alanine with α-ketoglutarate to indicate how this ping-pong reaction works.arrow_forwardName the following enzymes:a. enzyme responsible for the cutting of the sugar on the nonreducing ends of glycogen branches b. enzyme that is only present in the liver and kidney during glycogen utilization c. enzyme that catalyzes the transfer of a two-carbon fragment from a ketose donor to an aldose acceptor in PPParrow_forwardTransamination reactions have been described as ping-pong reactions. Use the reaction of alanine with - ketoglutarate to indicate how this ping-pong reaction works.arrow_forward
- In this transamination reaction (right), which of the following are the products X and Y? Oxaloacetate Glutamate A Alanine, a-ketoglutarate B Aspartate, a-ketoglutarate C Glutamate, alanine D Pyruvate, aspartatearrow_forwardUDP-glucuronosyltransferase enzymes bind the organic compound UDP-glucuronic acid (UDP-GA) in order to catalyse the transfer of a glucuronic acid group from UDP-GA to a drug molecule, releasing UDP from the active site as a product. UDP is then regenerated by the activity of another enzyme. What terms could be used to describe UDP-GA?arrow_forwardIdentify the following: a. An oxidative process that releases energy b. Cellular localization of the electron transport chain during cellular respiration. c. The number of double bonds present in the metabolic precursor of all prostaglandins.arrow_forward
- Regarding phosphofructokinase, which of the following statements is true: a. Low [ATP] stimulates the enzyme, but fructose-2,6-bisphosphate inhibits. b. High [ATP] stimulates the enzyme, but fructose-2,6-bisphosphate inhibits. c. The enzyme is more active at low [ATP] than at high, and fructose-2,6-bisphosphate activates the enzyme. d. High [ATP] stimulates the enzyme, and fructose-2,6-bisphosphate activates.arrow_forwardFive coenzymes are required by a-ketoglutarate dehydrogenase, the enzyme in the citric acid cycle that converts a-ketoglutarate to succinyl-CoA.a. Identify the coenzymes.b. Propose a mechanism for the reaction.arrow_forwardWhich of the following reactions is reversible and irreversible? Why? a. Pyruvate + β-hydroxybutyrate → lactate + acetoacetate b. Malate + pyruvate → oxaloacetate + lactate c. Acetaldehyde + succinate → ethanol + fumarate d. Pyruvate + NADH + H+ → lactate NAD+ e. Acetoacetate + NADH + H+ → β-hydroxybutyrate + NAD+ f. Malate + NAD+ → oxaloacetate + NADH + H+arrow_forward
- Five coenzymes are required by a-ketoglutarate dehydrogenase, the enzyme in the citric acid cycle that converts a-ketoglutarate to succinyl-CoA.a. Identify the coenzymes.b. Propose a mechanism for the reaction. A-ketoglutarate dehydrogenase A-ketoglutarate succinyl-CoAOO OO O−O O− −O SCoA + CO2arrow_forwardN-Acetylglutamate is required as a cofactor in the synthesis of carbamoyl phosphate. How is N-acetylglutamate synthesized from glutamate?arrow_forwardN -Acetylglutamate is required as a cofactor in the synthesis of carbamoyl phosphate. How is Nacetylglutamate synthesized from glutamate?arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





