
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 24.2, Problem 24.2PP
Interpretation Introduction
Interpretation:
The change in
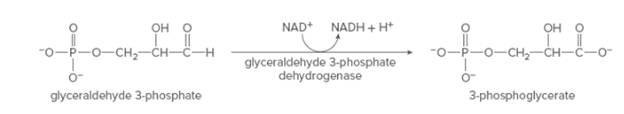 Concept Introduction:
Concept Introduction:
Aerobic respiration occurs in two steps;
- Glycolysis
- Citric acid cycle
Glycolysis is the first step that forms pyruvate. In the presence of oxygen means aerobic respiration, this pyruvate enters in the Krebs cycle and extracts energy in the form of electrons transfer. Electrons are transferred from the pyruvate to the receptors like
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
multiple choice, choose the correct answer
When substrate concentration is increased for a simple enzymatic reaction, the reaction order progresses from first-order to zero with respect to substrate concentration because: 1. S-S interactions at high concentrations become strong2. None of the enzyme is found in the ES complex3. Significant product formation results in inhibition of the reaction4. All of the active sites of the enzyme are saturated with substrate at high substrate concentration
(b) Determine how the reaction rate varies with substrate concentration.
Rate increases
substrate is added when enzyme is saturated
with substrate
Incorrect
Rate decreases
additional substrate is added when substrate
concentration is low
Answer Bank
Rate is unchanged
additional substrate is added when substrate
concentration is very high
<→CA https://session.masteringchemistry.com
Chapter 24 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 24.2 - Analyze the following reaction by considering the...Ch. 24.2 - Prob. 24.2PPCh. 24.3 - Prob. 24.1PCh. 24.3 - Prob. 24.2PCh. 24.3 - Prob. 24.3PCh. 24.3 - Prob. 24.4PCh. 24.3 - Prob. 24.5PCh. 24.3 - Prob. 24.6PCh. 24.4 - Prob. 24.7PCh. 24.4 - Prob. 24.8P
Ch. 24.4 - Prob. 24.9PCh. 24.5 - Prob. 24.10PCh. 24.5 - Prob. 24.11PCh. 24.5 - Prob. 24.12PCh. 24.6 - Prob. 24.13PCh. 24.7 - Prob. 24.14PCh. 24.7 - Prob. 24.3PPCh. 24.7 - Prob. 24.15PCh. 24.7 - Prob. 24.16PCh. 24.7 - Use the number of molecules of ATP formed from the...Ch. 24.7 - Prob. 24.18PCh. 24.8 - Prob. 24.19PCh. 24.8 - Prob. 24.20PCh. 24.8 - Prob. 24.21PCh. 24.9 - Prob. 24.4PPCh. 24.9 - What products are formed when each amino acid is...Ch. 24.9 - Prob. 24.22PCh. 24 - Analyze each reaction by considering the...Ch. 24 - Analyze each reaction by considering the...Ch. 24 - Prob. 25PCh. 24 - Prob. 26PCh. 24 - Prob. 27PCh. 24 - Prob. 28PCh. 24 - Prob. 29PCh. 24 - Prob. 30PCh. 24 - Prob. 31PCh. 24 - Prob. 32PCh. 24 - Glucose is completely metabolized to six molecules...Ch. 24 - Why is glycolysis described as an anaerobic...Ch. 24 - Write the overall equation with key coenzymes for...Ch. 24 - Prob. 36PCh. 24 - Prob. 37PCh. 24 - Prob. 38PCh. 24 - Consider the aerobic and anaerobic avenues of...Ch. 24 - Prob. 40PCh. 24 - Prob. 41PCh. 24 - Prob. 42PCh. 24 - Prob. 43PCh. 24 - Prob. 44PCh. 24 - Prob. 45PCh. 24 - Prob. 46PCh. 24 - Prob. 47PCh. 24 - Prob. 48PCh. 24 - Prob. 49PCh. 24 - Prob. 50PCh. 24 - Prob. 51PCh. 24 - Prob. 52PCh. 24 - Prob. 53PCh. 24 - Prob. 54PCh. 24 - Prob. 55PCh. 24 - Prob. 56PCh. 24 - Prob. 57PCh. 24 - Prob. 58PCh. 24 - Prob. 59PCh. 24 - How much ATP is generated by the complete...Ch. 24 - Prob. 61PCh. 24 - Fill in the boxes with the number of moles of each...Ch. 24 - Prob. 63PCh. 24 - Prob. 64PCh. 24 - Prob. 65PCh. 24 - Prob. 66PCh. 24 - Prob. 67PCh. 24 - Prob. 68PCh. 24 - Prob. 69PCh. 24 - Prob. 70PCh. 24 - What is the difference between ketogenic and...Ch. 24 - Prob. 72PCh. 24 - Prob. 73PCh. 24 - Draw the structure of the keto acid formed by the...Ch. 24 - Draw the products formed in each transamination...Ch. 24 - Prob. 76PCh. 24 - Prob. 77PCh. 24 - Prob. 78PCh. 24 - Prob. 79PCh. 24 - Prob. 80PCh. 24 - What metabolic intermediate is formed from the...Ch. 24 - What metabolic intermediate is formed from the...Ch. 24 - Prob. 83PCh. 24 - Prob. 84PCh. 24 - Prob. 85PCh. 24 - Prob. 86PCh. 24 - Prob. 87PCh. 24 - What is the cause of the pain and cramping in a...Ch. 24 - Prob. 89PCh. 24 - Prob. 90PCh. 24 - Prob. 91PCh. 24 - Prob. 92PCh. 24 - Prob. 93PCh. 24 - Prob. 94PCh. 24 - What type of enzyme would catalyze the conversion...Ch. 24 - Prob. 96PCh. 24 - Prob. 97CPCh. 24 - Prob. 98CPCh. 24 - Prob. 99CPCh. 24 - Prob. 100CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- - MULTIPLE CHOICE - Please answer properly QUESTION : Which of the following best describes the relationship between a reactant molecule and an enzyme? A. it is a permanent mutual alteration of structure B. it is one in which the enzyme is changed permanently C. it is an association stabilized by a covalent bond D. it is a temporary associationarrow_forward10. Factor In the enzyme catalyzed reaction depicted above, which factor could be changed (increased) to produce the graph? (Assume an unlimited substrate supply and change in the factor) a. inhibitor concentration Ob. temperature Oc. pH Od. enzyme concentrationarrow_forwardt Plase answer both of youarrow_forward
- Which of the following is TRUE? Check all that apply: O Enzymes decrease AG. Enzymes increase Ea. The velocity of a reaction is 28 mmol. The Km of an enzyme is 25 µmol/min. Enzymes decrease the energy of transition states.arrow_forwardAn enzyme has no activity at an extremely low pH. What could be an explanation for this observation? a. enzyme inhibition b. protein denaturation c. lack of cofactors d. substrate deactivation e. no correct response givenarrow_forwardMULTIPLE CHOICE - Please answer properly QUESTION : Supposed you used the enzyme lyase in the biomolecular laboratory, identify reaction that you have certainly observed with the enzyme Lyase. A. you observed intramolecular rearrangement of bonds B.you observed a transfer group from one molecule to another C. you observed breaking of bonds D. you observed formation of bondsarrow_forward
- Based on the chart shown, if there is unlimited enzyme potential with excess enzymes, why not always maintain excess enzymes in the body? Enzyme concentration (a) Reaction ratearrow_forward1. At what pH do the following enzymes work best? 1.2 Sucrase 0.8 0.6 0.4 0.2 10 15 pH a. Sucrase: b. Trypsin: Reaction Rate 186arrow_forward16.) Vmax represents the rate of reaction for an enzyme-substrate pairing under what conditions? B) when [S] = Km F) for all [S] Km C) when v Kmarrow_forward
- Consider the same enzyme catalyzed reaction with KM 25 mM, Vm 100 mM/s, and [S] =20 mM. A Noncompetitive inhibitor to this reaction has an inhibitor constant, K₁- 10 mM. What is the percent inhibition (%i) when the inhibitor concentration is [1]-5 mM? 50 33 67 25 Questionarrow_forwardplease post the correct answers and i will rate 49. Initial velocity is used to study enzyme kinetics because A The substrate concentration increases as the reaction proceeds B. The substrate concentration decreases as the reaction proceeds. C. The presence of inhibitor slows down the reaction. D. The presence of inhibitor stops the reaction. E. Cooperativity affects the reaction rate. 41. When myoglobin is compared to the beta chain of hemoglobin, A. The sequences and structures are all different. B. The sequences are essentially identical which is why the 3D structures are conserved. C. only 27 of more than 141 residues are identical, yet the 3D structures are conserved. D. only 27 of more than 141 residues are identical, which is why the 3D structures are not conserved. E. none of the above.arrow_forward5. In workshop and on the problem set we looked at a graph of rate of reaction vs. substrate concentration for an enzyme-catalyzed reaction in the presence of reversible inhibitors. On the graph below, draw a curve that shows what happens in the enzyme-catalyzed reaction in the presence of an irreversible inhibitor. Explain how you came up with your curve. You may assume the concentration of the inhibitor is less than the concentration of the enzyme. rate substrate concentrationarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY