
Concept explainers
(a)
Interpretation:
The number of acetyl CoA is formed from complete beta-oxidation of the oleic acid, needs to be identified.
Concept Introduction:
Beta − oxidation of fatty acids involves four series of reactions. In this reaction, the long-chain fatty acid is degraded into many two-carbon unit's acetyl CoA.
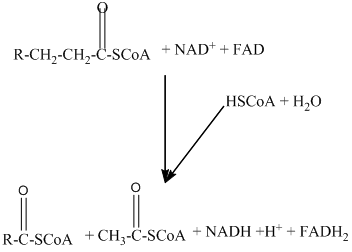
The number of acetyl CoA molecules formed, and number of time beta-oxidation occur can be determine by using the number of carbon atoms present in the fatty acid.
(b)
Interpretation:
The number of cycles of beta-oxidation is required for complete oxidation needs to be identified.
Concept Introduction:
Beta − oxidation of fatty acids involves four series of reactions. In this reaction, the long-chain fatty acid is degraded into many two-carbon unit's acetyl CoA.
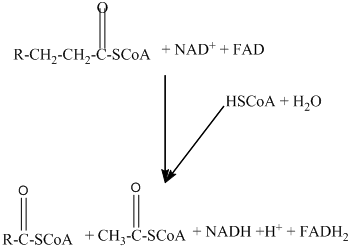
The number of acetyl CoA molecules formed, and the number of time beta-oxidation occur can be determined by using the number of carbon atoms present in the fatty acid.
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
General, Organic, and Biological Chemistry - 4th edition
- If an animal has 5.000 kg of palmitoyl-CoA available for fatty acid oxidation, how many kilograms of water could the animal produce from that palmitoyl-CoA? The chemical formula for palmitoyl-CoA is C37H66N7O17P3S.arrow_forwardWhich of the following are saponifiable lipids? a) triacylglycerols; f) stepoids; b) prostaglandins; g) glycolipids; c) waxes; h) terpenes; d) leukotrienes; i) phosphoglycerides; e) sphyngolipids; k) plasmalogens? Write the equation showing triacylglycerol formation from 1 mol of glycerol and 1 mol each of oleic, stearic and palmitic acids. Write the equation for the acid-catalyzed hydrolysis of the triacylglycerol: Write the equation for the saponification of the triacylglycerol: Write the equation for the hydrogenation of the triacylglycerol: Write the structure of the phosphoglyceride formed from 1 mol each of glycerol, stearic and palmitic acids, phosphoric acid, and choline . Show the formation of the sphingolipid containing oleic acid, phosphoric acid,…arrow_forwardStep 7 of the citric acid cycle is shown. Which statement best describes what occurs in this step? CO₂ 1 CH || CH + H₂O CO₂ fumarate CO₂™ fumarase HO C-H CH₂ CO₂ malate A) Fumarate undergoes hydrogenation with hydrogens and electrons provided by the enzyme fumarase. B) Fumarate undergoes hydration with the aid of the enzyme fumarase. C) Fumarate undergoes hydrolysis with the aid of the enzyme fumarase. D) Fumarate undergoes reduction with the aid of the cofactor fumarase.arrow_forward
- An oil obtained from coconuts is unusual in that all three fatty acid components are identical. The molecular formula of the oil is C45H86O6. What is the molecular formula of the carboxylate ion obtained when the oil is saponified?arrow_forwardFatty acids and triglycerides are an important source of nutrition and a dense form of stored energy. Digestion of fats yields more energy per gram than digestion of carbohydrates. Each turn on the B-oxidation spiral results in the formation of a new acetyl COA molecule. This molecule can then be used to generate more energy in A) fatty acid synthesis B) ketolysis C) glycolysis D) tricarboxylic acid cycle E) oxidative phosphorylationarrow_forwardWhy is CaCO3 the best catalyst for aspirin synthesis? What differentiates it from normal catalysts?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



