
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 23.3, Problem 10P
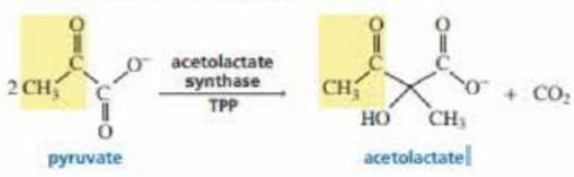
Acetolactate synthase transfers the acyl group of pyruvate to α-ketobutyrate. This is the first step in the biosynthesis of the amino acid isoleucine. Propose a mechanism for this reaction.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Acetolactate synthase transfers the acyl group of pyruvate to alpha-ketobutyrate. This is the first step in the biosynthesis of the amino acid isoleucine. Propose a mechanism for this reaction.
Cholesterol-lowering drugs commonly target
O HMG-COA synthase, the enzyme that catalyzes the synthesis of mevalonate
O pyruvate decarboxylase complex, which catalyzes the synthesis of acetyl-CoA
O mevalonate, an isoprene
O HMG-COAreductase, the rate-limiting enzyme in cholesterol biosynthesis
O HMG-CoA synthase, the enzyme that catalyzes the first step in cholesterol biosynthesis
The enzyme that catalyzes this reaction is a(n)
H
1000
CH3
Acetaldehyde
Otransferase
NADH + H+
ligase
Olyase
oxidoreductase
hydrolase
alcohol
dehydrogenase
NAD+
OH
CH2
CH3
Ethanol
Chapter 23 Solutions
Organic Chemistry (8th Edition)
Ch. 23.1 - Prob. 2PCh. 23.1 - Prob. 3PCh. 23.2 - How many conjugated double bonds are there in a....Ch. 23.2 - Instead of adding to the 4a position and...Ch. 23.2 - Prob. 7PCh. 23.3 - Prob. 8PCh. 23.3 - Acetolactate synthase is another TPP-requiring...Ch. 23.3 - Acetolactate synthase transfers the acyl group of...Ch. 23.3 - Prob. 12PCh. 23.5 - Which compound is more easily decarboxylated?
Ch. 23.5 - Prob. 14PCh. 23.5 - Explain why the ability of PLP to catalyze an...Ch. 23.5 - Explain why the ability of PLP to catalyze an...Ch. 23.5 - The enzyme that catalyzes the C C bond cleavage...Ch. 23.5 - Propose a mechanism for the ,-elimination reaction...Ch. 23.6 - Ethanolamine ammonia lyase, a coenzyme...Ch. 23.6 - Prob. 20PCh. 23.7 - How do the structure of tetrahydrofolate and...Ch. 23.7 - What is the source of the methyl group in...Ch. 23.8 - Thiols such as ethanethiol and propanethiol can be...Ch. 23 - How does the metal ion in carboxypeptidase A...Ch. 23 - Prob. 24PCh. 23 - Prob. 25PCh. 23 - For each of the following reactions, name both the...Ch. 23 - Prob. 27PCh. 23 - When transaminated, the three branched-chain amino...Ch. 23 - What acyl groups have we seen transferred by...Ch. 23 - Propose a mechanism for the following reaction:Ch. 23 - Draw the products of the following reaction, where...Ch. 23 - When UMP is dissolved in T2O, exchange of T for H...Ch. 23 - Dehydratase is a PLP-requiring enzyme that...Ch. 23 - In addition to the reaction mentioned in Section...Ch. 23 - PLP can catalyze both ,-elimination reactions...Ch. 23 - The glycine cleavage system is a group of four...Ch. 23 - Prob. 37PCh. 23 - FADH2 reduces , -unsaturated thioesters to...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Both norepinephrine and epinephrine are synthesized from the same protein-derived amino acid. From which amino acid are they synthesized? What types of reactions are involved in their biosynthesis?arrow_forwardOne of the steps in the pentose phosphate pathway for glucose catabolism is the reaction of sedoheptulose 7-phosphate with glyceraldehydes 3-phosphate in the presence of a transaldolase to yield erythrose 4-phosphate and fructose 6-phosphate. (a) The first part of the reaction is the formation of a protonated Schiff base of sedoheptulose 7-phosphate with a lysine residue in the enzyme followed by a retro-aldol cleavage to give an enamine plus erythrose 4-phosphate. Show the structure of the enamine and the mechanism by which it is formed. (b) The second part of the reaction is a nucleophilic addition of the enamine to glyceraldehyde 3-phosphate followed by hydrolysis of the Schiff base to give fructose 6-phosphate. Show the mechanism.arrow_forwardDiazepam is converted in vivo to an active metabolite Oxazepam. By using chemical equations show metabolic transformation of diazepam to oxazepam. Provide name of enzymes involved in these reactions.arrow_forward
- The enzyme catalysts for which gene expression levels are increased during gluconeogenesis in the liver are: pyruvate carboxylase, aldolase, and phosphofructokinase glucose-6-phosphatase, fructose-1,6-bisphosphatase, pyruvate carboxylase, and phosphoenolpyruvate carboxykinase hexokinase, phosphofructokinase 1, phosphofructokinase 2, and pyruvate kinase glucose-6-kinase, aldolase, and enolasearrow_forwardIn the body, during the citric acid cycle the following reaction occurs as the first part of an enzyme mediated process. но OH YYYYYY OH OH H₂O NaBH4 LiAlH4 H₂SO4 and heat K₂Cr2O7 HQ OH What reagent would be used to bring about this reaction in the laboratory? OH 3arrow_forward4. Liver alcohol dehydrogenase (LADH) catalyzes a reversible, pH-dependent oxidation of an alcohol to an aldehyde according to the following reaction: LADH RCH2OH + NAD* RCHO + NADH + H* where NAD* and NADH are, respectively, the oxidized and reduced forms of the cofactor nicotin-amide adenine dinucleotide. While the reaction is actually a two-substrate reaction, it proceeds like a one- substrate reaction through a so-called compulsory-ordered ternary complex mechanism. It has been shown through kinetic studies that, in the case of primary alcohols, the enzyme binds first the cofactor forming a binary enzyme-NAD* complex that then binds the second substrate, the alcohol, to form a ternary enzyme-NAD*-substrate complex in which the oxidation of the alcohol proceeds with reduction of the cofactor. While the enzyme catalyzes oxidation of various steroids as well as ingested ethanol, it also catalyzes the oxidation of methanol CH3OH, forming the extremely toxic product formaldehyde HCHO.…arrow_forward
- The rate-limiting step is a metabolic pathway is the slowest step which determines the overall rate of the other reactions in the pathway. In glycolysis, the rate limiting step is a phosphorylation reaction where phosphofructokinase (PFK-1) catalyzes the reaction fructose-6-bisphosphate -> fructose-1,6-bisphosphate, the same step in gluconeogenesis. Select one: The statement is FALSE. The statement is TRUE.arrow_forwardIf an enzyme-catalyzed reaction has a high rate at low pH and low rate at higher pH, this implies that a group on either the enzyme or the substrate must be for an efficient reaction. leaving group oxidoreductase coenzymes O protonated deprotonated The compound that consists of deoxyribose linked by an N-glycosidic bond to N-9 of guanine is: adenylate deoxyguanosine guanosine nucleotide guanylatearrow_forwardDraw the reaction catalyzed by the enzyme: Carbonic anhydrase.arrow_forward
- What acyl group does pyruvate decarboxylase transfer to a proton?arrow_forwardPlease draw by hand. Triosephosphate isomerase (TIM) catalyzes the conversion of dihydroxyacetone phosphate to glyceraldehyde-3-phosphate. The enzyme's catalytic groups are Glu 165 and His 95. In the first step of the reaction, these catalytic groups function as a base and an acid catalyst, respectively. Propose a mechanism for the reaction. ОН 2-03Р0 ОН dihydroxyacetone phosphate triosephosphate isomerase 2-03РО. H glyceraldehyde-3-phosphate FYI Glu is glutamic acid and his is histadinearrow_forwardKinetic versus thermodynamic. The reaction of NADH with oxygen to produce NAD+NAD* and H2OH2O is very exergonic, yet the reaction of NADH and oxygen takes place very slowly. Why does a thermodynamically favorable reaction not occur rapidly?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY