
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 23, Problem 15P
How would you transform tetradecanal into each of the following?
(a)
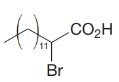
(b)
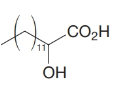
(c)
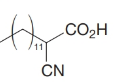
(d)
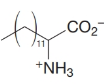
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Explain why each of the following alkenes is stable or unstable.(a) 1,2-dimethylcyclopentene (b) trans-1,2-dimethylcyclopentene(c) trans-3,4-dimethylcyclopentene (d) trans-1,2-dimethylcyclodecene
Draw the structure of the following compounds all showing C and H atoms.(a) 2-methyl -3-iso propyl heptanes(b) Dicyclopropyl methane.
6) Which is the organic product for the following reaction?
(a)
(b)
(c)
(d)
сон
COOH
ОН
ОН
COOH
COOH
KMnO4
H2O
Chapter 23 Solutions
Organic Chemistry
Ch. 23 - Prob. 1PPCh. 23 - Prob. 2PPCh. 23 - Prob. 3PPCh. 23 - Prob. 4PPCh. 23 - Prob. 5PPCh. 23 - Prob. 6PPCh. 23 - Prob. 7PPCh. 23 - Prob. 8PPCh. 23 - Prob. 9PPCh. 23 - Prob. 10PP
Ch. 23 - Prob. 11PPCh. 23 - Prob. 12PPCh. 23 - Prob. 13PPCh. 23 - Prob. 14PCh. 23 - 23.15 How would you transform tetradecanal into...Ch. 23 - Prob. 16PCh. 23 - Prob. 17PCh. 23 - When limonene (Section 23.3) is heated strongly,...Ch. 23 - Gadoleic acid (C20H38O2), a fatty acid that can be...Ch. 23 - 23.20 -Phellandrene and -phellandrene are isomeric...Ch. 23 - Prob. 21PCh. 23 - Prob. 22PCh. 23 - Prob. 23PCh. 23 - The initial steps of a laboratory synthesis of...Ch. 23 - Prob. 25PCh. 23 - Prob. 26PCh. 23 - Prob. 27PCh. 23 - 2. The biosynthesis of fatty acids is accomplished...Ch. 23 - Prob. 3LGPCh. 23 - Prob. 4LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
18.23 Do the reactions involved in ozone depletion involve changes in oxidation state of the O atoms? Explain.
Chemistry: The Central Science (13th Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Write the equilibrium law for each of the following heterogeneous reactions. (a)2Hg(l)+Cl2(g)Hg2Cl2(s) (b)Disso...
Chemistry: The Molecular Nature of Matter
1.6 Read the labels on products used to wash your dishes. What are the names of some chemicals contained in tho...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Consider conditions for which a fluid with a free stream velocity of V=1m/s flows over a surface with a charact...
Fundamentals of Heat and Mass Transfer
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Starting from bromoethane, the formation of which of the following compound requires more than one step of reaction? 2 (a) Methoxyethane (b) Ethanol (c) Ethanoic acid (d) Ethenearrow_forward6) Which is the organic product for the following reaction? (a) (b) (c) (d) LOH OH COOH OH OH COOH COOH KMnO4 H₂O (e) None of the above products will be formedarrow_forward1B. Provide the IUPAC name of the following compounds, with clear indication of stereochemistry for stereocenters and alkene. Me. (a) (b) (c) (d) Br "Ме CIarrow_forward
- 1 Provide the IUPAC name of the following compounds, with clear indication of stereochemistry for stereocenters and alkene. (a) (b) Br (c) Me. (d) Mearrow_forwardDraw the structures of the following molecules: (a) (S)-sec-butylcyclopentane (b) p-benzyltoluene c) (Z)-5-phenyl-3-methyl-3-octenearrow_forwardIf either of the following molecules has a stereogenic carbon (chirality center), give structures for both of the enantiomers. (a) 2-bromopropane (b) 2-bromobutanearrow_forward
- Provide the IUPAC name for each of the following compounds. Pay close attention to stereochemistry. (a) (b) -NH2 (c) OH ОН NH2 NH2 НО .CI Br. NH2 O,N"arrow_forwardThe IUPAC name of the compound is (a) 5-formylhex-2-en-3-one (b) 5-methyl-4-oxohex-2-en-5-al (c) 3-keto-2-methylhex-5-enal (d) 3-keto-2-methylhex-4-enal The correct statement regarding electrophile is (a) electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from another electrophile (b) electrophiles are generally neutral species and can form a bond by accepting a pair of electrons from a nucleophile (c) electrophile can be either neutral or positively charged species and can form a bond by accepting a pair of electrons from a nucleophile (d) electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from a nucleophile, Which among the given molecules can exhibit tautomerism? Ph Ph I III (a) III only (b) Both I and III (d) Both II and III (c) Both I and II 5) Which of the following biphenyls is optically active? IIarrow_forwardProvide the IUPAC name for each of the following compounds. Pay attention to stereochemistry. (c) (a) (Б) О NO2 OH NH2 ОН NH2arrow_forward
- Answer ALL parts of this question. (a) (b) What type of reagent is shown in Figure Q26a? CH3MgBr Figure Q26a (e) Fermentation of sugar can be used to produce impure ethanol. What extra practical step would then be required to isolate purified ethanol? (c) What functional group (i.e. class of compounds) is produced when aldehydes are treated with sodium borohydride? (d) (i)Draw the structure of the major product formed when propene (Figure Q26d) undergoes a hydration reaction (with water), and name it according to the IUPAC nomenclature. Propene Figure Q26d (ii) Explain why this product in part (i) is the Markovnikov product. Why are alcohols less acidic compared to phenols? Draw diagrams to explain your reasoning.arrow_forwardWhich is least reactive in electrophilic aromatic substitution? (A) (B) CH3 (C) CHO (D) C3arrow_forwardFor each pair of compounds, predict which compound has a higher boiling point. then explain why that compound has a higher boiling point. (a) isopropyl bromide and n-butyl bromide (b) isopropyl chloride and tert-butyl bromide (c) 1-bromobutane and 1-chlorobutanearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License