
Rank the following molecule in order of the increasing solubility in water.
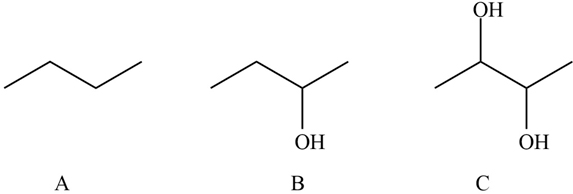
The ranking of the molecules in increasing order of their solubility in water.
Answer to Problem 3TC
Solubility of C molecule is highest then B and A has least solubility. Increasing order of solubility in water is
Explanation of Solution
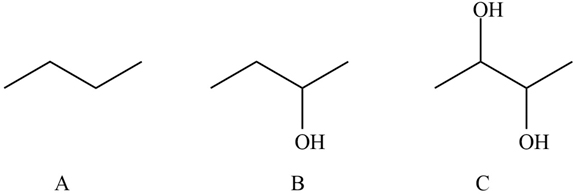
A) There are no OH group present in the molecule hence there will be no formation of the H-bond between the water molecule and butane molecule. Hence,it is insoluble.
B) Here OH group is present but the number OH group present in C are more than that of B so even though it will form H-bonding with water molecule the solubility is less than that of C molecule.
C) The number of OH group is highest so the solubility of C molecule is highest as more H-bonding formed with water is highest.
Conclusion:
Therefore, Solubility of C molecule is highest then B and A has least solubility. Increasing order of solubility in water is
Chapter 22 Solutions
Conceptual Physical Science Explorations
Additional Science Textbook Solutions
University Physics (14th Edition)
Conceptual Physical Science (6th Edition)
Cosmic Perspective Fundamentals
Conceptual Physics (12th Edition)
The Cosmic Perspective Fundamentals (2nd Edition)
Tutorials in Introductory Physics
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





