
Concept explainers
(a)
Interpretation:
With the help of given mass values, infrared and
Concept Introduction:
Mass spectrum: It generates multiple ions from the sample under investigation, it than separates them according to their specific mass to charge ration
The
IR Spectrum: It is concerned with the study of absorption of infrared, which causes vibration transition in the molecule. It is mainly used to identify
(b)
Interpretation:
The apperance of peaks in the mass spectrum of compound C at the given
Concept introduction:
Basic principle of mass spectrum: It generates multiple ions from the sample under investigation, it than separates them according to their specific mass to charge ration
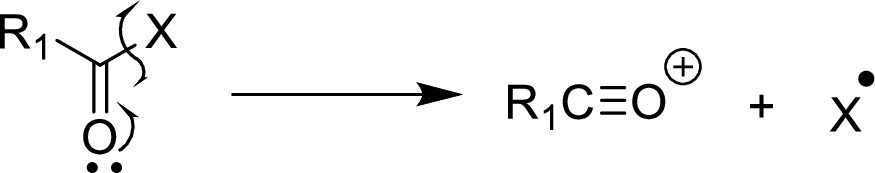
Alpha
It is an expected pathway for carbonyl compounds, ethers, halide, alcohols and
Trending nowThis is a popular solution!

Chapter 21 Solutions
Organic Chemistry
- Compound B has molecular formula C9H10. The IR spectrum is shown below. The 1H-NMR spectrum shows a multiplet at 7.2 ppm integrating to 4H, a triplet at 2.9 ppm integrating to 4H, and a triplet at 2.1 ppm integrating to 2 H. Suggest a structure for B and explain your reasoningarrow_forwardCompound A has molecular formula C7H7X. Its 1H-NMR spectrum shows a singlet at 2.25 ppm and two doublets, one at 7.28 ppm and one at 7.39 ppm. The singlet has an integral of three and the doublets each have an integral of two. The mass spectrum of A shows a peak at m/z = 126 and another peak at m/z = 128; the relative height of the two peaks is 3:1 respectively. - Identify what atom X is, explaining your reasoning - Identify Compound A, explaining your reasoningarrow_forwardCompound P has molecular formula C5H9ClO2. Deduce the structure of P from its 1H and 13C NMR spectra.arrow_forward
- Following are IR and 1H-NMR spectra of compound D. The mass spectrum of compound D shows a molecular ion peak at m/z 136, a base peak at m/z 107, and other prominent peaks at m/z 118 and 59. Q.) Propose structural formulas for ions in the mass spectrum at m/z 118, 107, and 59.arrow_forwardTreatment of alcohol A (molecular formula C5H12O) with CrO3, H2SO4, and H2O affords B with molecular formula C5H10O, which gives an IR absorption at 1718 cm−1. The 1H NMR spectrum of B contains the following signals: 1.10 (doublet, 6 H), 2.14 (singlet, 3 H), and 2.58 (septet, 1 H) ppm. What are the structures of A and B?arrow_forwardA and B are isomeric dicarbonyl compounds of the molecular formula C5H&O2. The 'H NMR spectrum of A contains a singlet at 2.05 ppm and another singlet at 5.40 ppm. The 'H NMR spectrum of B contains three signals: a singlet at 2.3 ppm, a triplet at 1.10 ppm and a quartet at 2.70 ppm. Suggest structures for A and B and draw them in their respective boxes below. 1st attemptarrow_forward
- The 1H-NMR spectrum of compound R, C6H14O, consists of two signals: d 1.1 (doublet) and d 3.6 (septet) in the ratio 6:1. Propose a structural formula for compound R consistent with this informationarrow_forward(a) Compound A has molecular formula C9H18O but shows only one singlet in the 1H-NMR spectrum. Suggest a structure for A and explain your reasoning. (b) Compound B has molecular formula C10H14. The IR, mass, 1H-NMR, and 13C-NMR speca are shown below, they are also downloadable for closer inspection by clicking the link under the spectral data. Suggest a structure for B and explain your reasoning.arrow_forwardTwo compounds with the molecular formula C5H10O have the following 1H and 13C NMR data. Both compounds have a strong IR absorption band in the 1710–1740-cm_1 region. Elucidate the structure of these two compounds and interpret the spectra. (10) (a) 1H NMR: d 2.55 (septet, 1H), 2.10 (singlet, 3H), 1.05 (doublet, 6H); 13C NMR: d 212.6, 41.5, 27.2, 17.8 (b) 1H NMR: d 2.38 (triplet, 2H), 2.10 (singlet, 3H), 1.57 (sextet, 2H), 0.88 (triplet, 3H); 13C NMR: d 209.0, 45.5, 29.5, 17.0, 13.2arrow_forward
- Compound C shows a molecular ion at m/z 148 and other prominent peaks at m/z 105 and 77. Following are its infrared and 1H-NMR spectra. Q.) Deduce the structural formula of compound Carrow_forwardCompounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra.Compound A: δ 1.3 (3H, triplet); 3.6 (2H, quartet); 4.1 (2H, singlet); 11.1 (1H, broad singlet)Compound B: δ 2.6 (2H, triplet); 3.4 (3H, singlet); 3.7 (2H triplet); 11.3 (1H, broad singlet)arrow_forwardThe mass, IR, and 1H-NMR spectra of a compound are shown below. Suggest a possible structure for the compound, explaining how you reach your decision.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





