
Concept explainers
Which of the molecules and ions given in Problem 21.15 are aromatic according to the Hückel criteria? Which, if planar, would be antiaromatic?
21.15 State the number of 2p orbital electrons in each molecule or ion.
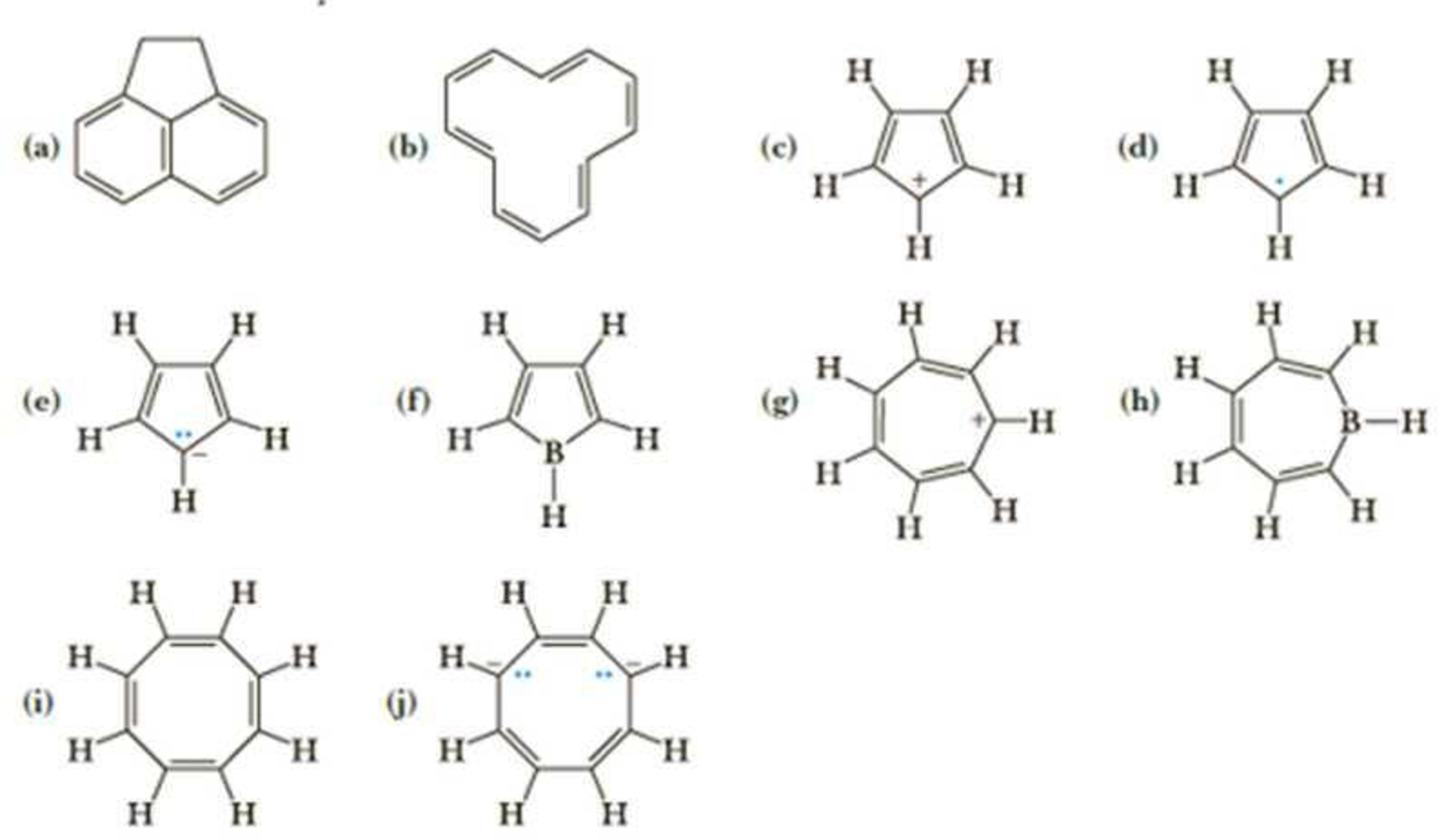
(a)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
There are five
Huckel’s rule of aromaticity
(b)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
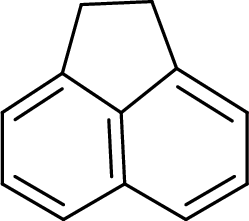
The given compound is shown here:
There are six
Huckel’s rule of aromaticity
The given compound is planar. So, it is expected to possess aromaticity due to the expected effective delocalization of
(c)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
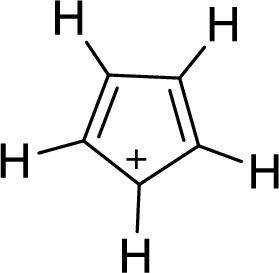
There are two
Huckel’s rule of aromaticity
The given compound is planar. So, it is expected to possess aromaticity due to the expected effective delocalization of
(d)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
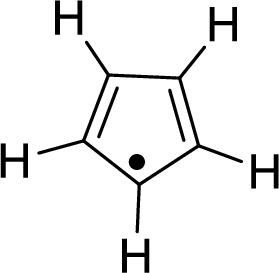
There are two
Huckel’s rule of aromaticity
(e)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
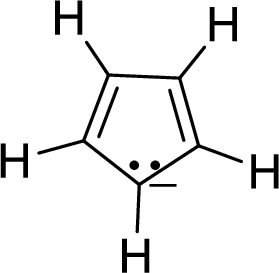
There are two
Huckel’s rule of aromaticity
(f)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
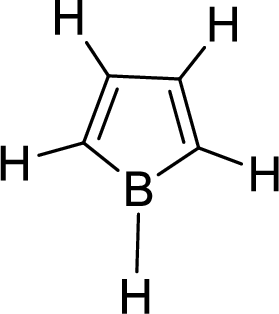
There are two
Huckel’s rule of aromaticity
The given compound is planar. So, it is expected to possess aromaticity due to the expected effective delocalization of
(g)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
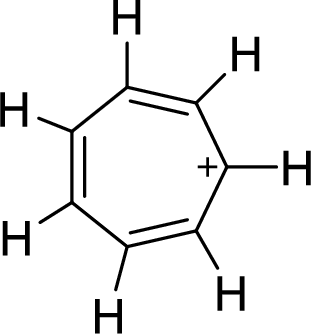
There are three
Huckel’s rule of aromaticity
(h)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
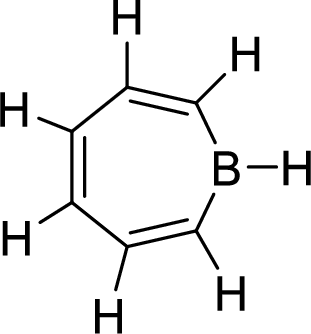
There are three
Huckel’s rule of aromaticity
(i)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
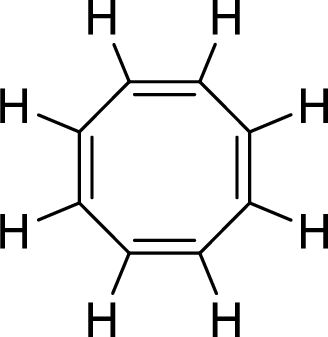
There are four
Huckel’s rule of aromaticity
(j)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
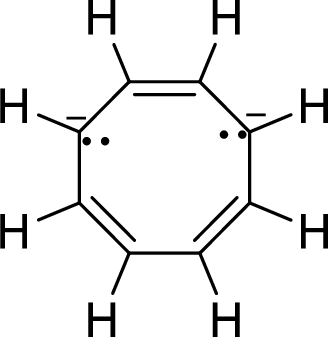
There are three
Huckel’s rule of aromaticity
Want to see more full solutions like this?
Chapter 21 Solutions
Organic Chemistry
- Is the molecule aromatic, antiaromatic, or non-aromatic? Why?arrow_forwardAromatic heterocyclic chemistry: State whether the following drugs are aromatic heterocycles and if they follow the 4 aromaticity rules (unsaturation, (4n + 2)pi electrons, cyclic and flat/planar), NAME ANY HETEROCYCLES. Draw the structure of the core of any aromatic heterocycles in the drug. Riociguat, Imatinib, Clopidogrel, Glatiramer acetate, Axitinibarrow_forwardBiphenyl has the following structure.(a) Is biphenyl a (fused) polynuclear aromatic hydrocarbon?(b) How many pi electrons are there in the two aromatic rings of biphenyl? How does this number compare with that for naphthalene?(c) The heat of hydrogenation for biphenyl is about 418 kJ>mol (100 kcal>mol). Calculate theresonance energy of biphenyl.(d) Compare the resonance energy of biphenyl with that of naphthalene and with that of two benzene rings. Explain thedifference in the resonance energies of naphthalene and biphenyl.arrow_forward
- To be aromatic, a molecule must have 4n + 2 πs electrons and must have a planar, monocyclic system of conjugation. Cyclodecapentaene fulfills one of these criteria but not the other and has resisted all attempts at synthesis. Explain.arrow_forwardIs this molecule aromatic? Why doesn't the pi bond in the benzene ring on the left side not count torward aromaticity?arrow_forwardAromatic heterocyclic chemistry: State whether the following drugs are aromatic heterocycles and if they follow the 4 aromaticity rules (unsaturation, (4n + 2)pi electrons, cyclic and flat/planar), NAME ANY HETEROCYCLES Vildagliptin, Valbenazine, Levetiracetam, (Dolutegravir/Lamivudine)arrow_forward
- 1 i. What is Resonance Theory? Sate five conclusions that can be drawn from the theory. ii. State the two main experiments that were used to establish the extra stability of the benzene molecule. iii. What are the factors that confer Aromaticity to an organic molecule? iv. State the effects of substituents on a benzene derivative towards further aromatic substitution. V. Based on the above suggest the various types of substituents that can be attached to Benzene.arrow_forwardDefine Inscribed Polygon Method for Predicting Aromaticity ?arrow_forwardProve the aromaticity of the molecules given below by discussing them.arrow_forward
- Cyclopropenones are described as having aromatic character. How would you account for this, given that the ring contains three π-electrons?arrow_forwardDiscuss the aromaticity of the below compounds? Explain which compound is aromatic, why?arrow_forwardHow many pi electrons are there in the two aromatic rings of biphenyl?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


