
Interpretation: The diene and the dienophile that might be used to prepare the given racemic Diels-Alder adduct have to be found.
Concept Introduction:
Diels-Alder reaction:
It is the reaction of conjugated dienes with double or triple bonded compounds which are known as “dienophiles”. The reaction is a
Example:
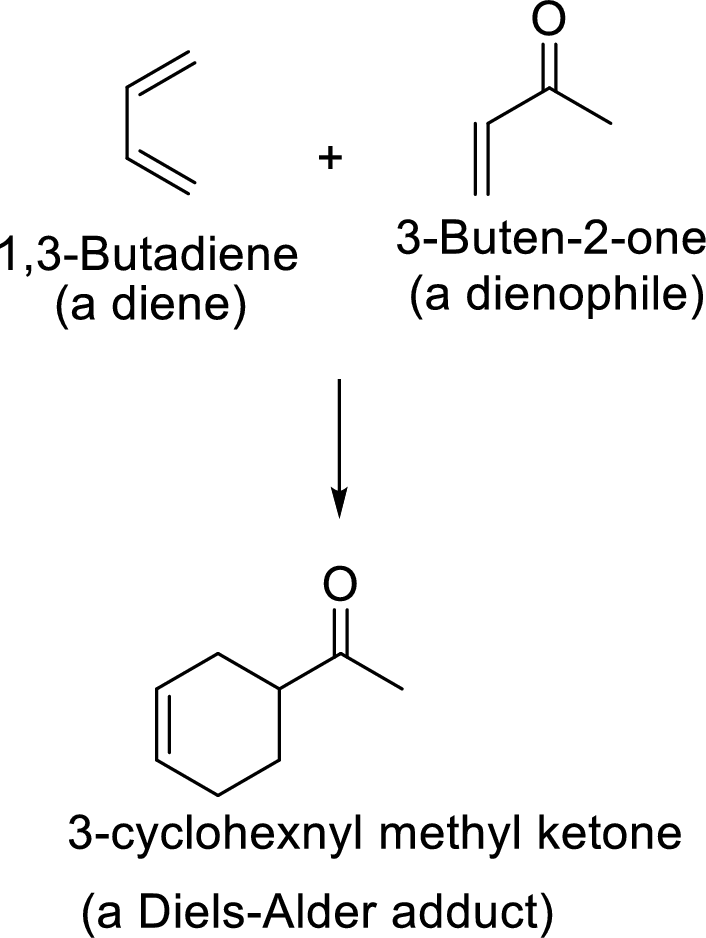
Diels-Alder reaction to form bicyclic system:
The Diels-Alder adduct formed in the Diels-Alder reaction can also be a bicyclic system which will be obtained when cylopentadiene is used as the diene as shown here:
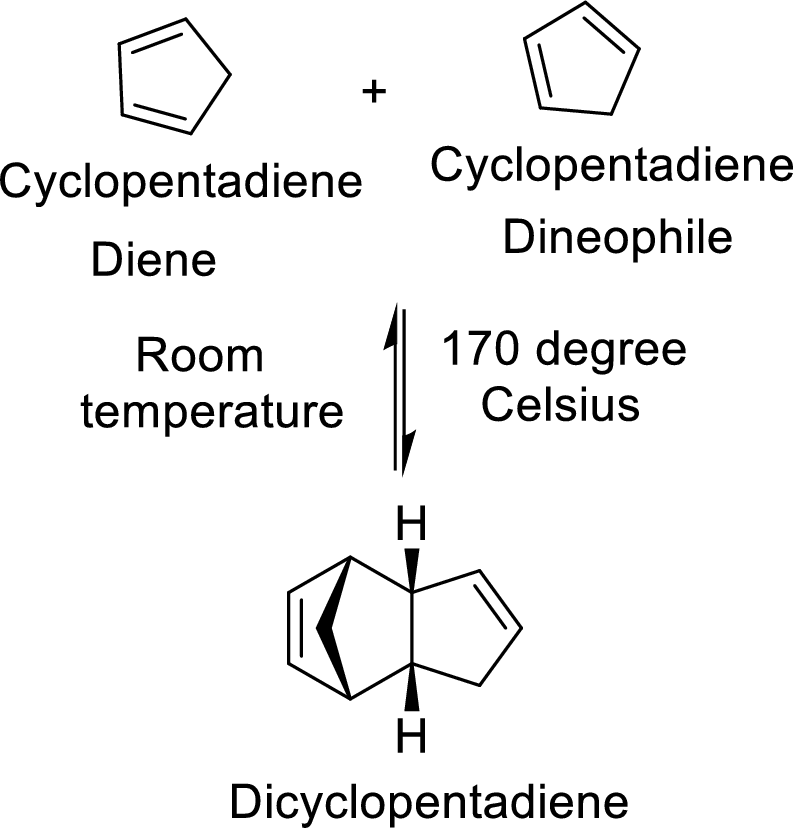
In this reaction, the cylopentadiene acts as both diene and dienophile and formed a bicyclic system. When it is heated to
Racemic mixture in Diels-alder reaction:
In the Diels-alder reaction, the formation of two new sigma bonds results in the formation of two new chiral centres that are enantiomers to each other. So, the Diels-alder adduct is the mixture of two enantiomers and therefore it is being a racemic Diels-alder adduct.
Trending nowThis is a popular solution!

Chapter 20 Solutions
Organic Chemistry
- Following is an example of a type of reaction known as a Diels-Alder reaction 1,3-Pentadiene Ethylene 3-Methylcyclohexene (a racemic mixture) The Diels-Alder reaction between a diene and an alkene is quite remarkable in that it is one of the few ways that chemists have to form two new carbon-carbon bonds in a single reaction. Given what you know about the relative strengths of carbon-carbon sigma and pi bonds, would you predict the Diels-Alder reaction to be exothermic or endothermic? Explain your reasoning.arrow_forwardWhat reactants are expected to afford the following Diels-Alder adduct? H. H;C H. 請arrow_forward4 predict the diels alder starting materialarrow_forward
- The following triene undergoes an intramolecular Diels-Alder reaction to give a bicyclic product. Propose a structural formula for the product. Account for the observation that the Diels-Alder reaction given in this problem takes place under milder conditions (at lower temperature) than the analogous Diels-Alder reaction shown in Problem 20.34.arrow_forwardWhat diene and dienophile would react to form the following Diels-Alder product. COH CO2Harrow_forwardWhat diene and dienophile would react to form the following Diels-Alder product. CO₂H CO₂Harrow_forward
- The Diels-Alder reaction is not limited to making six-membered rings with only carbon atoms. Predict the products of the following reaction that produce rings with atoms other than carbon in them.arrow_forward3) How could you use Diels-Alder reactions to prepare the following products? Show the starting diene and dienophile in each case H a. b. C. H H d. H .CO₂CH3 CNarrow_forwardPredict the regiochemical outcome (major product) for the following Diels-Alder reaction: MeO CHO ? MeO MeO OHC MeO O MeO. + O Meo, CHO CHO CHO CHO хсно - сно CHO Осно CHO CHOarrow_forward
- 4. Predict the product for the following Diels-Alder reaction.arrow_forward9. For the following Diels-Alder products, propose the correct starting materials needed. D. a. b. C. fo{- ●●● Xox -arrow_forwardCan someone draw the arrows for this reaction so I can understand how the product was formed? I'm having trouble understanding that aspect of the Diels-Alder reaction.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

