
Interpretation:
Whether an achiral or two equal enantiomeric products would be given has to be predicted in the product of the given reaction and explained with chair like transition state.
Concept Introduction:
Cope-rearrangement:
It is a pericyclic reaction that involves the redistribution of six electrons through the formation of a cyclic transition state from which a
Example with mechanism of cope-arrangement:
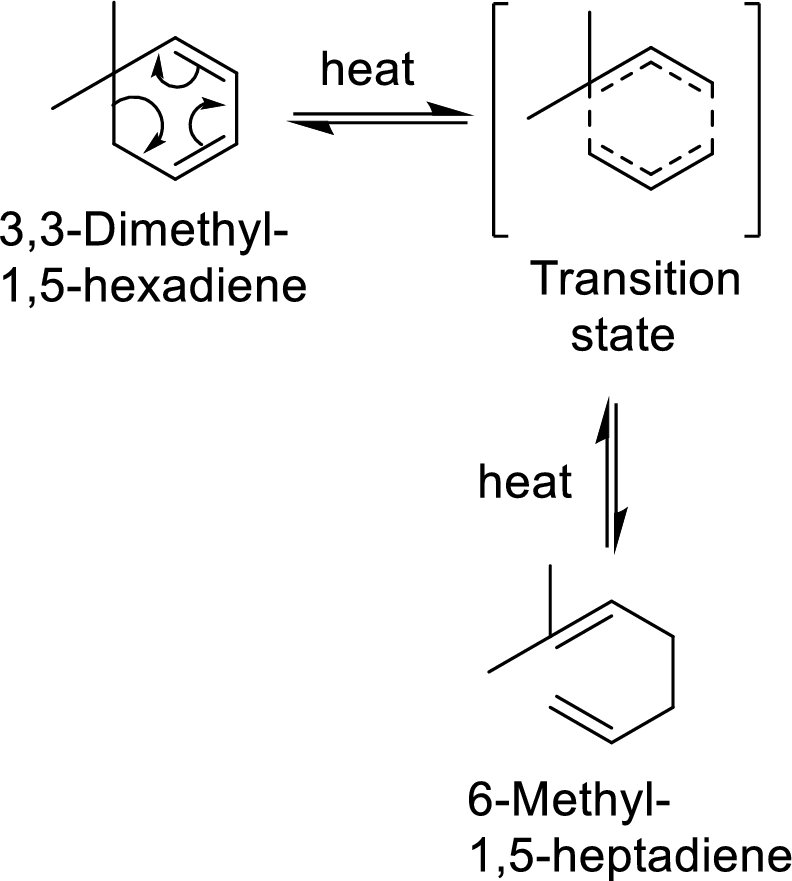
In this mechanism, two pi-bonds and one sigma bond of the reactant molecule has been rearranged and formed two new pi-bonds through a cyclic transition state.
Identification of cope-rearrangement in a
In the cope-rearrangement, the flow of electrons takes place between six bonds that are bonded as
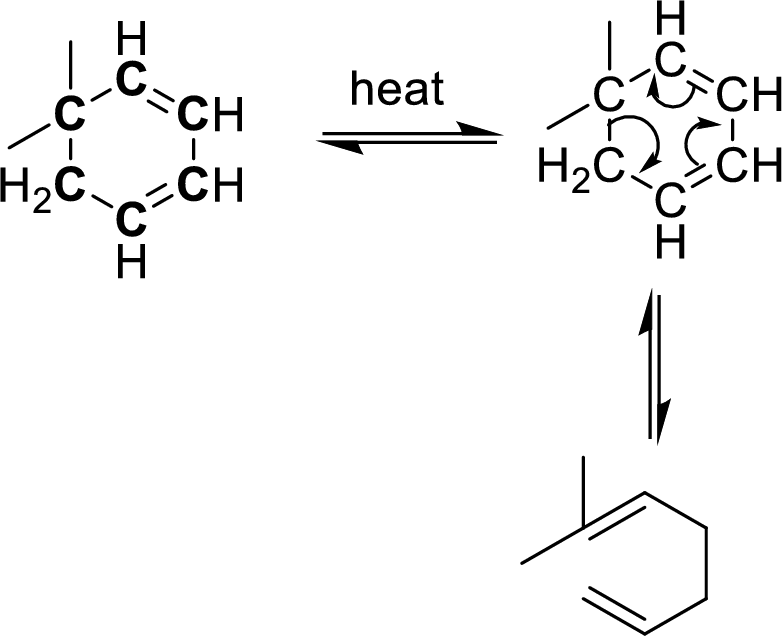
The carbon atoms that are involving in the cope-rearrangement are shown in bold.
Stereochemistry in a product formed:
- • In the product of a
chemical reaction , if a carbon atom has been attached with four different carbon atoms, then it is known as chiral carbon atom or stereocenter in the product. - • The bonds of the
functional groups because of which a new chiral carbon is supposed to form have to be represented in solid wedge bond and hashed-wedge bonds according to the particular enantiomer. - • Racemic mixture is the mixture of two enantiomers in equal proportions.
- • Enantiomers are non-superimposable mirror images.
- • Achiral product is the product in which there won’t be any chiral centre or stereocenter.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Organic Chemistry
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardWhen 2,2-dimethylcyclohexanol is treated with acid, 1,2-dimethylcyclohexene and iso- propylidenecyclopentane are the products obtained. Draw a detailed mechanism that explains this result. CH3 H3O+ CH3 CH3 CH3 OH CH3 CH3 2,2-dimethyl- cyclohexanol 1,2-dimethyl- isopropylidene- cyclopentane cyclohexenearrow_forwardPredict the major product of the following reaction. If a single enantiomer will be obtained, be sure to draw it with well-defined stereochemistry. If a mixture of enantiomers will be obtained, either draw both enantiomers or label the product as "racemic".arrow_forward
- When cyclohexene is reacted with hydrobromic acid in acetic acid, the major product is bromocyclohexane. There is a small amount of cyclohexyl acetate formed. What is the mechanism that forms both compounds? What is the purification procedure that isolates both compounds?arrow_forwardThe reaction of (S)-2-bromopentane with potassium cyanide to yield 2-methylpentanenitrile (2-cyanopentane) occurs via a nucleophilic substitution pathway. The reaction is 100% stereospecific.arrow_forwardWhen trans-2-chloro-1-cyclohexanol is treated with a base, cyclohexene oxide is the product. However, when cis-2-chloro-1-cyclohexanol is treated with a base, the product is cyclohexanone. Write the mechanism for each of the two reactions.arrow_forward
- Draw reaction mechanisms with all reactants, arrows, intermediates, and products. Your mechanism must account for all the products if more than one product is formed. 4-methycyclohexanol with phosphoric acid H3PO4 to for 1-methycyclohexene, 3- methylcyclohexene and 4-methycyclohexenearrow_forwardAs we will learn in Chapter 9, an epoxide is an ether with an oxygen atom in a three-membered ring. Epoxides can be made by intramolecular SN2 reactions of intermediates that contain a nucleophile and a leaving group on adjacent carbons, as shown.Assume that each of the following starting materials can be converted to an epoxide by this reaction. Draw the product formed (including stereochemistry) from each starting material. Why might some of these reactions be more difficult than others in yielding nucleophilic substitution products?arrow_forwardThe major product of the given reaction has the molecular formula C10H16O3. Draw its structure in the most stable tautomeric form.arrow_forward
- The following molecule can undergo keto–enol tautomerization upon addition of hydroxide. Draw the structure of the enol form and the mechanism by which it forms.arrow_forwardFrom what you have learned about enols and the hydration of alkynes, predict what product is formed by the acidcatalyzed hydration of CH3CH2CH2C = COCH3. Draw a stepwise mechanism that illustrates how it is formed.arrow_forwardComplete the following reactions by providing the correct product(s), starting materials, or reagents. When appropriate, products should have the correct stereochemistryarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

