
Concept explainers
(a)
Interpretation:
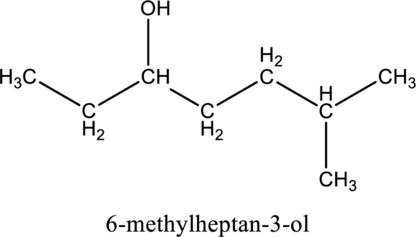
The systematic name and given alcohol should be given and it should be classified as primary, secondary or tertiary.
Concept introduction:
IUAC gives rules for the naming of chemical compounds. These rules are,
For saturated alcohols, longest chain with hydroxyl group is the parent chain of the compound and remaining groups and atoms are consider as substituent. In systematic name parent chain is ended with ol suffix.
In the nomenclature, Find the parent chin first then number the carbon atoms in the parent chain by giving lowest number to the hydroxyl substituted carbon followed by designate the other substituent in its position in parent chain.
If more than one same substituent or hydroxyl groups were occurs, add a prefix (di-, tri-, tetra-, ect..) in front of parent chain name.
Finally the systematic name was written as, the substituent position number with prefix (di-, tri-, tetra-, ect..) of number of substituents followed by name of the substituent then write the parent chain name (which contains large number of carbon atoms in the chain).
Alcohols:
In
In primary alcohols, hydroxyl group is connected with carbon, which is connected with all hydrogen or connected with single carbon substituent.
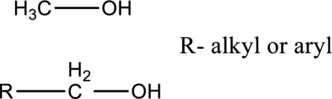
In secondary alcohols, the hydroxyl group is connected with carbon, which is connected two alkyl or aryl substituents.
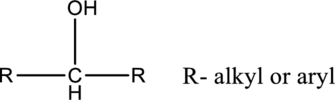
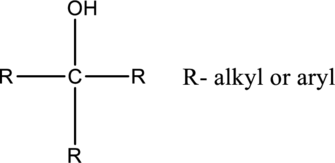
In tertiary alcohols, the hydroxyl group is connected with carbon, which is connected two alkyl or aryl substituents.
(b)
Interpretation:
The systematic name and given alcohol should be given and it should be classified as primary, secondary or tertiary.
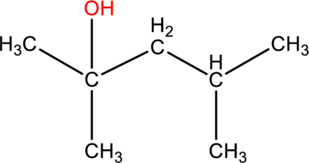
Concept introduction:
Refer part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Foundations of College Chemistry 15e Binder Ready Version + WileyPLUS Registration Card
- Give the classification of the following alcohols as to primary, secondary, or tertiary.arrow_forwardClassify alkyl halides as primary, secondary, tertiary, aliphatic, or benzenearrow_forwardDistinguish between a primary, a secondary and tertiary alcohol by using an example with three (3) isomers of butanol. Label each one.arrow_forward
- identify such alcohol is primary, secondary or tertiaryarrow_forwardExplain the concept of Cyclohexane Conformations ?arrow_forward(a) Enter the IUPAC name for this alcohol: CH,CH,CHCH, ОН Classify it as a primary, secondary, or tertiary alcohol: (b) Enter the IUPAC name for this alcohol: CH,CHCH,CH,CHOH CH, ČH, Classify it as a primary, secondary, or tertiary alcohol:arrow_forward
- Given the molecular formula C5H12O, give the structures (any form) and names of a primary, secondary, and tertiary alcohol, and an ether (ether can be named for EC).arrow_forwardDifferentiate primary, secondary, and tertiary alcohols.arrow_forwardClassify alcohols as primary, secondary, or tertiaryarrow_forward
- are they: secondary, primary, tertiary or phenol alcohol?arrow_forwardWhat are some examples of primary, secondary, and tertiary alcohols?arrow_forward2. Write bond-line structural formulas for (a.) two primary alcohols, (b.) a secondary alcohol, and (c.) atertiary alcohol—all having the molecular formula C4H10O.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





