
(a)
Interpretation: The overall balance
Concept Introduction:
In a chemical reaction to be occurred; two most important requirements are activation energy to reactant molecule and correct orientation of reactant molecules to colloid and form product.
(a)
Answer to Problem 129CP
Explanation of Solution
On the basis of number of steps in a chemical reaction, the reactions can be classified as elementary reactions and multi-step reaction. The elementary step of a chemical reaction can be defined as the primary step of the reaction which comprise together to show the complete conversion of a reactant to product in the multi-step reaction.
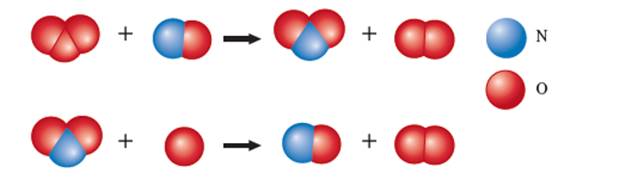
The given ball and stick model for the chemical reaction is:
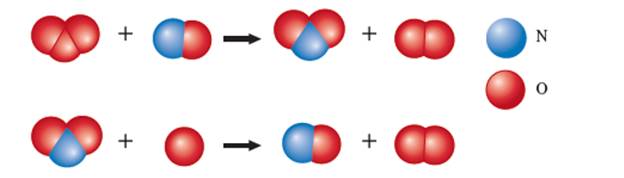
Hence the steps of the given reaction can be written as:
(b)
Interpretation: The species which act as catalyst for the ozone destruction needs to be determined.
Concept Introduction:
Chemical kinetic is the branch of chemistry that deals with kinetic or rate of a chemical reaction. The rate of reaction can be defined as the change in the concentration of reactant and product with time. The rate of reaction mainly depends on the activation energy.
In a chemical reaction to be occurred; two most important requirements are activation energy to reactant molecule and correct orientation of reactant molecules to colloid and form product.
(b)
Answer to Problem 129CP
NO must be catalyst for the given reaction.
Explanation of Solution
Catalyst can be defined as the substance which enhance the rate of reaction and again regenerate at the end of reaction.
In the given reaction NO involves in step-1 and again regenerate in step-2 therefore it is not part of overall reaction. Hence NO must be catalyst for the given reaction.
(c)
Interpretation: The species which act as intermediate for the ozone destruction needs to be determined.
Concept Introduction:
Chemical kinetic is the branch of chemistry that deals with kinetic or rate of a chemical reaction. The rate of reaction can be defined as the change in the concentration of reactant and product with time. The rate of reaction mainly depends on the activation energy.
In a chemical reaction to be occurred; two most important requirements are activation energy to reactant molecule and correct orientation of reactant molecules to colloid and form product.
(c)
Answer to Problem 129CP
Explanation of Solution
The species which is formed during the reaction and will convert to product at the end of reaction is called as intermediate.
In the given reaction
(d)
Interpretation: The rate law for the given ozone destruction reaction if the step-1 is slow and step-2 is fast step needs to be determined.
Concept Introduction:
Chemical kinetic is the branch of chemistry that deals with kinetic or rate of a chemical reaction. The rate of reaction can be defined as the change in the concentration of reactant and product with time. The rate of reaction mainly depends on the activation energy.
In a chemical reaction to be occurred; two most important requirements are activation energy to reactant molecule and correct orientation of reactant molecules to colloid and form product.
(d)
Answer to Problem 129CP
Explanation of Solution
Given:
In the given reaction, the step-1 is slow step and step-2 is fast step therefore the rate law will be determined with the help of step-1. In step-1; O3 and NO take part therefore the rate law can be written as:
(e)
Interpretation: The two step mechanism with second step and overall balance reaction for the rate law for ozone destruction by chlorine which are formed by reaction of Freons with light needs to be determined.
Concept Introduction:
Chemical kinetic is the branch of chemistry that deals with kinetic or rate of a chemical reaction. The rate of reaction can be defined as the change in the concentration of reactant and product with time. The rate of reaction mainly depends on the activation energy.
In a chemical reaction to be occurred; two most important requirements are activation energy to reactant molecule and correct orientation of reactant molecules to colloid and form product.
(e)
Answer to Problem 129CP
Explanation of Solution
The destruction of ozone with NO is given below:
Freons can form active Cl atoms in the presence of light which can further react with ozone to form oxygen gas. The mechanism can be written as:
Want to see more full solutions like this?
Chapter 18 Solutions
Chemical Principles
- 1. The amount of carbon dioxide in the Earth's early atmosphere decreased because it was used by plants and algae for photosynthesis, dissolved in the oceans and formed fossil fuels. Give one other way that the amount of carbon dioxide in the Earth's early atmosphere decreased,arrow_forward[Thermodynamics] In normal chemical storage, the element sulfur remains unreacted with oxygen in the atmosphere. However, it is impossible for sulfur and oxygen to react spontaneously to form gaseous sulfur dioxide. This is most likely to happen: A. rusting of iron B. combustion of methane C. at very low temperatures D. only in the presence of a catalyst E. oxidation of glucosearrow_forwardORGANIC CHEMISTRY. What substance is used in the determination of Chlorine? a. Copper Oxide b. Soda lime c. Sucrosearrow_forward
- Solid gold (III) oxide undergoes a thermal decomposition reaction to form gold metal and oxygen gas. 1. Write the balanced equation for this reaction, including physical states. 2. This reaction will go to completion without the need for a catalyst like MnO2 with potassium chlorate. Based on this information, which element, gold or chlorine, more readily is reduced in these reactions? Explain. 3. When a student was completing this experiment, the atmospheric pressure was measured to be 751 torr. The temperature of the water was found to be 23.0°C. What is the pressure (in torr) of the oxygen gas, PO2, that is produced?arrow_forward5.Why is potassium nitrate also in airbags? 6. Write the balanced equation for the reaction between potassium nitrate and sodium below.arrow_forwardWarming of the atmosphere is due to the greenhouse effect. What is responsible for this phenomenon? a. The CO2 level in the atmosphere is increased b. The global temperature is decreased tremendously c. There is a widespread destruction on the green color of plants. d. The amount of oxygen in the atmosphere is greatly affected.arrow_forward
- Out of white phosphorus and red phosphorus, which one is more reactive and why?arrow_forward15. Carbon dioxide makes up approximately 0.04% of Earth's atmosphere. If you collect a 2.0-L sample from the atmosphere at sea level (101.33 kPa) on a warm day at 27 °C, how many CO2 molecules are in your sample? * a. 5 x 10^20 b. 5 x 10^21 O c. 5x 10^22 d. 5 x 10^23arrow_forwardWhat exact color does ozone gas have? and explain why it got that colorarrow_forward
- What are the main sources of halogen-containing compounds? Why is it important to know their concentration in the atmosphere? Use of chemical reactions are welcomedarrow_forwardSome chemical reactions are sensitive to air (oxygen or moisture), they are conducted under nitrogen atmosphere. Why? d.arrow_forwardWhich of the following process add carbon to the atmosphere? 1. Carbonic acid formation and limestone formation. 2. Photosynthesis and carbonic acid formation. 3. Photosynthesis and limestone formation. 4. Cellular respiration and fossil fuel combustion.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





