
Concept explainers
(a)
Interpretation: The hybridization of central atom in
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(a)
Answer to Problem 119AE
Explanation of Solution
To draw the Lewis structure of
Number of valence electrons in S = 6
Number of valence electrons in F = 7
Total number of valence electrons in
Hence the Lewis structure of
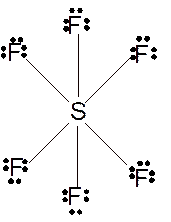
The hybridization can be checked with the help of Lewis structure and VSEPR theory which provide the geometry of molecules with lone pairs.
(b)
Interpretation: Interpret the hybridization of central atom in
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(b)
Answer to Problem 119AE
Explanation of Solution
To draw the Lewis structure of
Number of valence electrons in Cl = 7
Number of valence electrons in F = 7
Total number of valence electrons in
Hence the Lewis structure of
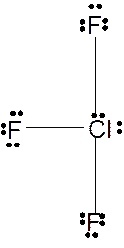
The hybridization can be checked with the help of Lewis structure and VSEPR theory which provide the geometry of molecules with lone pairs.
(c)
Interpretation: The hybridization of central atom in
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(c)
Answer to Problem 119AE
Explanation of Solution
To draw the Lewis structure of
Number of valence electrons in Ge = 4
Number of valence electrons in Cl = 7
Total number of valence electrons in
Hence the Lewis structure of
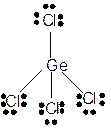
The hybridization can be checked with the help of Lewis structure and VSEPR theory which provide the geometry of molecules with lone pairs.
(d)
Interpretation: The hybridization of central atom in
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The shape or geometry of molecule can be predicted with the help of hybridization and VSEPR theory. It can be checked with the below formula:
(d)
Answer to Problem 119AE
Explanation of Solution
To draw the Lewis structure of
Number of valence electrons in Xe = 8
Number of valence electrons in F = 7
Total number of valence electrons in
Hence the Lewis structure of
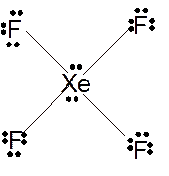
The hybridization can be checked with the help of Lewis structure and VSEPR theory which provide the geometry of molecules with lone pairs.
Want to see more full solutions like this?
Chapter 18 Solutions
Chemical Principles
- What modification to the molecular orbital model was made from the experimental evidence that B2 is paramagnetic?arrow_forwardSketch the resonance structures for the N2O molecule. Is the hybridization of the N atoms the same or different in each structure? Describe the orbitals involved in bond formation by the central N atom.arrow_forwardSketch the resonance structures for the nitrate ion, NO3. Is the hybridization of the N atom the same or different in each structure? Describe the orbitals involved in bond formation by the central N atom.arrow_forward
- What hybridization is required for central atoms exhibiting trigonal bipyramidal geometry? Octahedral geometry? Describe the bonding of PF5, SF4, SF6, and IF5, using the localized electron model.arrow_forwarda Nitrogen trifluoride, NF3, is a relatively unreactive, colorless gas. How would you describe the bonding in the NF3 molecule in terms of valence bond theory? Use hybrid orbitals. b Silicon tetrafluoride, SiF4, is a colorless gas formed when hydrofluoric acid attacks silica (SiO2) or glass. Describe the bonding in the SiF4 molecule, using valence bond theory.arrow_forwardGive the expected hybridization of the central atom for the molecules in Exercises 21 and 22.arrow_forward
- What hybridization is required for central atoms that have a tetrahedral arrangement of electron pairs? A trigonal planar arrangement of electron pairs? A linear arrangement of e lectron pairs? How many unhybridized p atomic orbitals are present when a central atom exhibits tetrahedral geometry? Trigonal planar geometry? Linear geometry? What are the unhybridized p atomic orbitals used for? Describe the bonding in H2S, CH4, H2CO, and HCN, using the localized electron model.arrow_forwardIn addition to NF3, two other fluoro derivatives of nitrogen are known: N2F4 and N2F2. What shapes do you predict for these two molecules? What is the hybridization for the nitrogen in each molecule?arrow_forwardA model of the organic compound allene is shown below. (a) Explain why the allene molecule is not flat. That is. explain why the CH2 groups at opposite ends do not lie in the same plane. (b) What is the hybridization of each of the carbon atoms in allene? (c) What orbitals overlap to form the bonds between carbon atoms in allene?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





