
(a)
Interpretation: The products formed for the given Diels-Alder reaction are to be drawn.
Concept introduction: Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene. The dienophile adds to one side of the diene, and diene adds to the one side of the dienophile. Thus, they have syn stereochemistry.
Answer to Problem 16.50P
The products formed by the given Diels-Alder reaction are shown below.
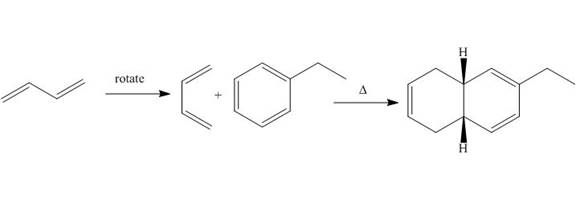
Explanation of Solution
The given reaction is shown below.
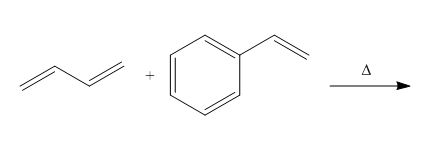
Figure 1
Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene. The dienophile adds to one side of the diene, and diene adds to the one side of the dienophile. Thus, they have syn stereochemistry.
The products formed by the given Diels-Alder reaction are shown below
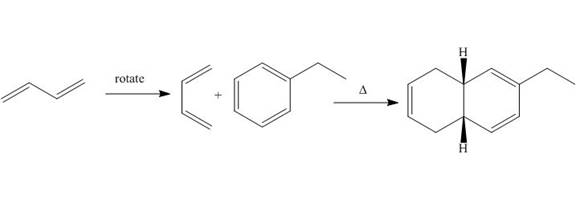
Figure 2
The products formed for the given Diels-Alder reaction is shown in Figure 2.
(b)
Interpretation: The products formed for the given Diels-Alder reaction are to be drawn.
Concept introduction: Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene. The dienophile adds to one side of the diene, and diene adds to the one side of the dienophile. Thus, they have syn stereochemistry.
Answer to Problem 16.50P
The products formed by the given Diels-Alder reaction are shown below.
Explanation of Solution
Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene. The dienophile adds to one side of the diene, and diene adds to the one side of the dienophile. Thus, they have syn stereochemistry.
The products formed by the given Diels-Alder reaction are shown below
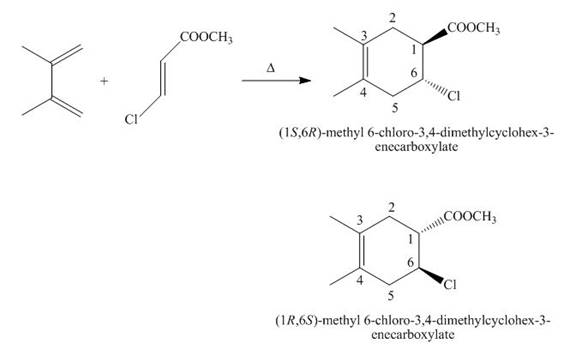
Figure 4
The products formed for the given Diels-Alder reaction is shown in Figure 4.
(c)
Interpretation: The products formed for the given Diels-Alder reaction are to be drawn.
Concept introduction: Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene.
Answer to Problem 16.50P
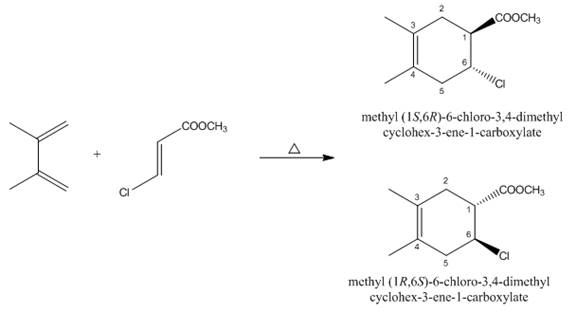
The products formed by the given Diels-Alder reaction are shown below.
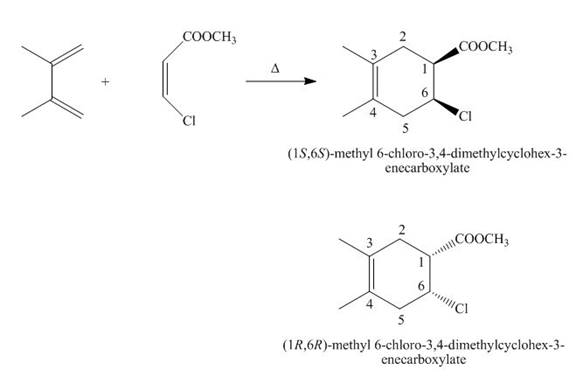
Explanation of Solution
The given reaction is shown below.
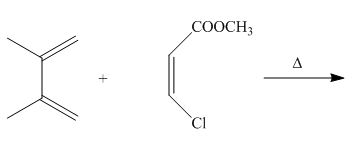
Figure 5
Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene. The dienophile adds to one side of the diene, and diene adds to the one side of the dienophile. Thus, they have syn stereochemistry.
Therefore, the product formed for the given Diels-Alder reaction is shown below.

Figure 6
The product formed for the given Diels-Alder reaction is shown in Figure 6.
(d)
Interpretation: The product formed for the given Diels-Alder reaction is to be drawn.
Concept introduction: Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene. The dienophile adds to one side of the diene, and diene adds to the one side of the dienophile. Thus, they have syn stereochemistry.
Answer to Problem 16.50P
The product formed for the given Diels-Alder reaction is shown below.
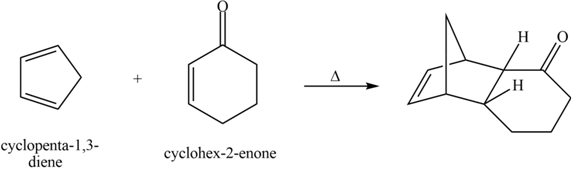
Explanation of Solution
The given reaction is shown below.
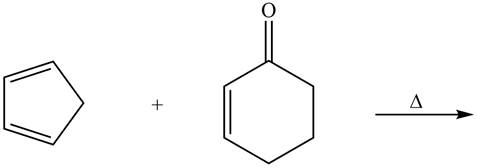
Figure 7
Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene. The dienophile adds to one side of the diene, and diene adds to the one side of the dienophile. Thus, they have syn stereochemistry.
Therefore, the product formed for the given Diels-Alder reaction is shown below.
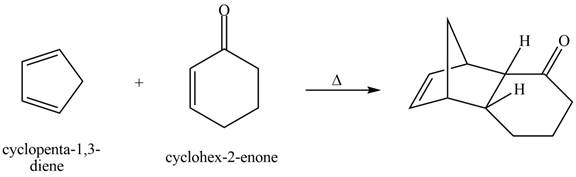
Figure 8
The product formed for the given Diels-Alder reaction is shown in Figure 8.
(e)
Interpretation: The product formed for the given Diels-Alder reaction is to be drawn.
Concept introduction: Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene. The dienophile adds to one side of the diene, and diene adds to the one side of the dienophile. Thus, they have syn stereochemistry.
Answer to Problem 16.50P
The product formed for the given Diels-Alder reaction is shown below.
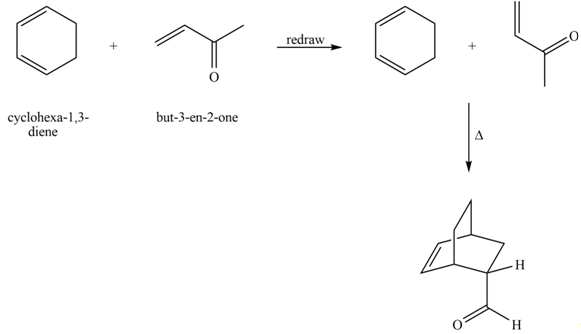
Explanation of Solution
The given reaction is shown below.
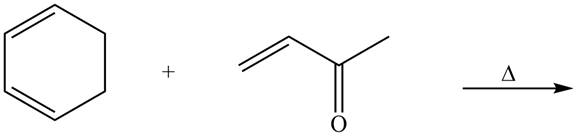
Figure 9
Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene. The dienophile adds to one side of the diene, and diene adds to the one side of the dienophile. Thus, they have syn stereochemistry.
The product formed for the given Diels-Alder reaction is shown below.
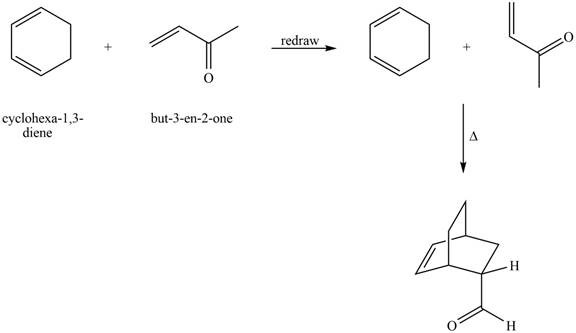
Figure 10
The products formed for the given Diels-Alder reaction are shown in Figure 10.
(f)
Interpretation: The product formed for the given Diels-Alder reaction is to be drawn.
Concept introduction: Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene. The dienophile adds to one side of the diene, and diene adds to the one side of the dienophile. Thus, they have syn stereochemistry.
Answer to Problem 16.50P
The product formed for the given Diels-Alder reaction is shown below.
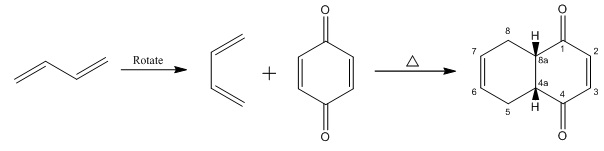
Explanation of Solution
The given reaction is shown below.
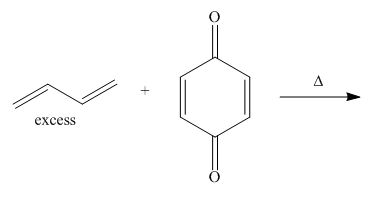
Figure 11
Diels-alder reaction is a cycloaddition reaction in which two molecules combine to form a new ring. In this type of reaction, syn addition takes place. It is a reaction between diene with a dienophile to yield a cyclohexene. The dienophile adds to one side of the diene, and diene adds to the one side of the dienophile. Thus, they have syn stereochemistry.
The product formed for the given Diels-Alder reaction is shown below.
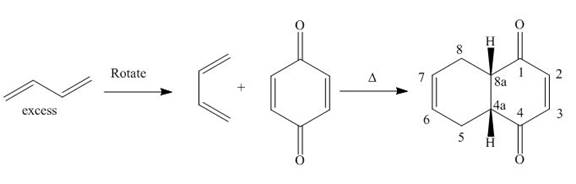
Figure 12
The products formed for the given Diels-Alder reaction are shown in Figure 12.
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry
- What diene and dienophiles are used to make the following compounds? of a. b.arrow_forwardPlease show all arrow pushingj mechanisais. Thank you!arrow_forwardWhat name reaction would result to the formation of the cyclohexene structure below? R .CO̟Et A. Diels-Alder reaction B. Wittig Reaction C. Friedel-Craft reaction D. Claisen Condensationarrow_forward
- Electrophilic Addition Soubong neblA-aleid rose 9160910 31 babeen ene singo 14.43 Draw the products formed when each compound is treated with one equivalent of HBr. a. b. C.arrow_forwardWhat type of sigmatropic rearrangement is illustrated in each equation? a. b. Darrow_forwardDevise a stepwise synthesis of attached compound from dicyclopentadieneusing a Diels–Alder reaction as one step. You may also use organiccompounds having ≤ 4 C's, and any required organic or inorganicreagents.arrow_forward
- Rank the following alkenes from most to least stable. A. В. С. D. B.arrow_forwardWhich of the structures below is the major product obtained from the following Diels-Alder reaction. Note that you will have to re-draw the diene in the s-cis conformation to correctly predict the stereochemistry of the product. ? A B Carrow_forwardDraw the product of each Diels-Alder reaction and indicate the stereochemistry at all stereogenic centers. A a. b.arrow_forward
- Draw the product formed when the following diene and dienophile react in a Diels-Alder reaction. draw structure ..arrow_forwardDevise a stepwise synthesis of attached compound from dicyclopentadiene using a Diels–Alder reaction as one step. You may also use organic compounds having ≤ 4 C's, and any required organic or inorganicreagents.arrow_forwardA Moving to another question will save this response. Question 20 For a diene to undergo a Diels-Alder reaction it must: O A. be able to adopt cyclohexane O B. be substituted with electron-withdrawing groups. Oc. be able to adopt and s-trans conformation. O D. be able to adopt an s-cis conformation. O E. be substituted with electron-donating groups. Moving to another question will save this response. O Type here to searcharrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





