
(a)
Interpretation:
The product formed by the reaction of given diene with one equivalent of
Concept introduction:
Diene is a hydrocarbon that contains two
Answer to Problem 16.15P
The product formed by the reaction of given diene with one equivalent of
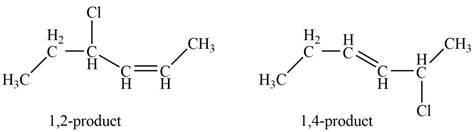
Explanation of Solution
Conjugated dienes undergoes electrophilic addition to gives a mixture of products that is
Markovnikov addition of
The given diene is shown below.

Figure 1
The given diene is a conjugated diene. The attack of
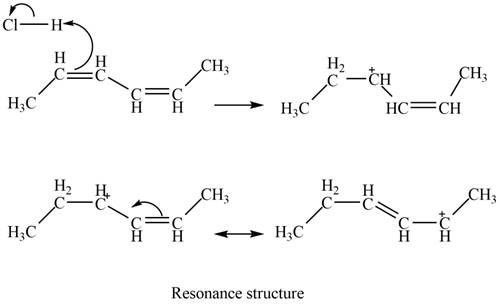
Figure 2
In the next step, chlorine as a nucleophile will attack on the carbocation to give constitutional isomers. Thus, the product formed by the reaction of given diene with one equivalent of
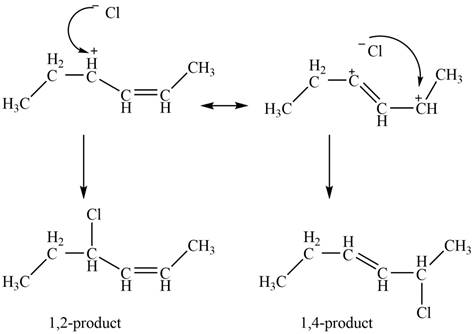
Figure 3
The product formed by the reaction of given diene with one equivalent of
(b)
Interpretation:
The product formed by the reaction of given diene with one equivalent of
Concept introduction:
Diene is a hydrocarbon that contains two
Answer to Problem 16.15P
The product formed by the reaction of given diene with one equivalent of
Explanation of Solution
Conjugated dienes undergoes electrophilic addition to gives a mixture of products that is
Markovnikov’s rule states that the positive part of acid attached to that carbon atom in
The given diene is shown below.
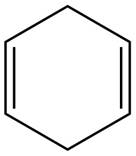
Figure 4
The given diene is an isolated diene. The attack of

Figure 5
In the next step, chlorine as a nucleophile will attack on the carbocation to form desired product. Thus, the product formed by the reaction of given diene with one equivalent of
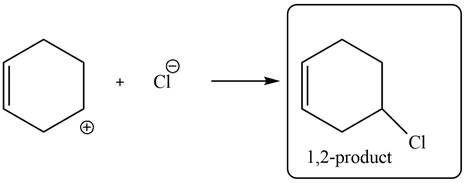
Figure 6
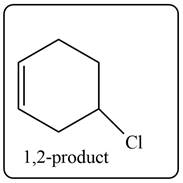
The product formed by the reaction of given diene with one equivalent of
(c)
Interpretation:
The product formed by the reaction of given diene with one equivalent of
Concept introduction:
Diene is a hydrocarbon that contains two
Answer to Problem 16.15P
The product formed by the reaction of given diene with one equivalent of
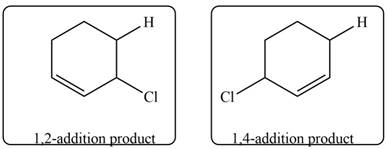
Explanation of Solution
Conjugated dienes undergoes electrophilic addition to gives a mixture of products that is
Markovnikov addition of
The given diene is shown below.
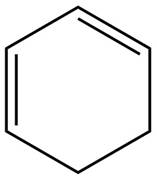
Figure 7
The given diene is a conjugated diene. The attack of
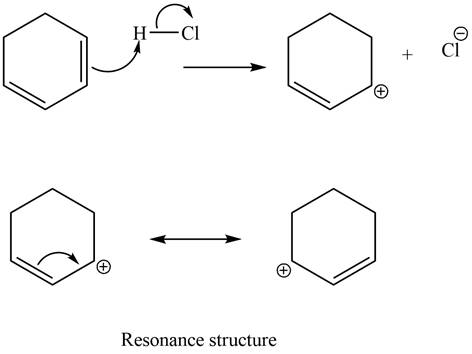
Figure 8
In the next step, chlorine as a nucleophile will attack on the carbocation to give constitutional isomers. Thus, the product formed by the reaction of given diene with one equivalent of
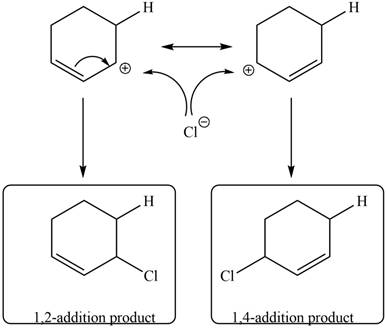
Figure 9
The product formed by the reaction of given diene with one equivalent of
(d)
Interpretation:
The product formed by the reaction of given diene with one equivalent of
Concept introduction:
Diene is a hydrocarbon that contains two
Answer to Problem 16.15P
The product formed by the reaction of given diene with one equivalent of
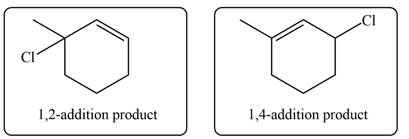
Explanation of Solution
Conjugated dienes undergoes electrophilic addition to gives a mixture of products that is
Markovnikov addition of
The given diene is shown below.
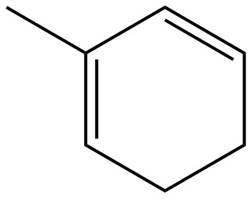
Figure 10
The given diene is a conjugated diene. The attack of
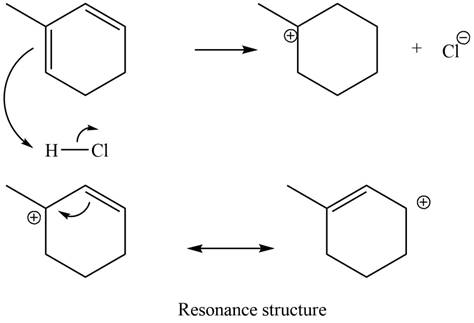
Figure 11
In the next step, chlorine as a nucleophile will attack on the carbocation to give constitutional isomers. Thus, the product formed by the reaction of given diene with one equivalent of
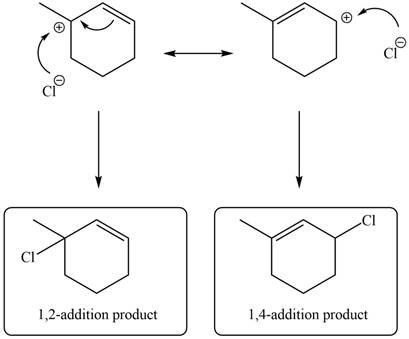
Figure 12
The product formed by the reaction of given diene with one equivalent of
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry
- Draw the product formed when each diene and dienophile react in a Diels–Alder reaction.arrow_forwardDraw the products formed when each alkene is treated with O3 followed by Zn, H2O.arrow_forwardDraw the products formed when a-D-gulose is treated with each reagent.a. CH3I, Ag2Ob. CH3OH, HClc. Ac2O, pyridined. The product in (a), then H3O+e. The product in (b), then Ac2O, pyridinef. The product in (d), then C6H5CH2Cl, Ag2Oarrow_forward
- Draw the products formed when hex-1-yne is treated with each reagent. Show arrow pushing as well. [1] R2BH; [2] H2O2 , HO−arrow_forwardFill in any starting materials, reagents, or products to complete the reaction.arrow_forwardDraw the products (including stereoisomers) formed in each reaction.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
