
Cyclobutadiene and (Cyclobutadiene)tricarbonyliron
As we saw in Section
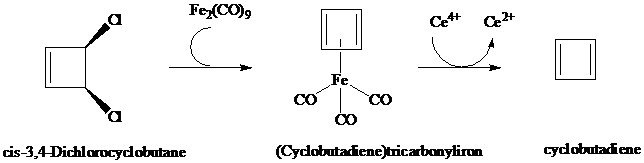
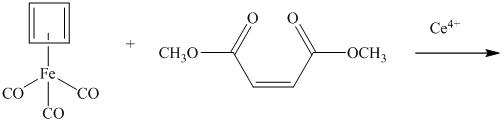
What is the product of the following reaction?

Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry - Standalone book
- Deuterium (D, or 2H) is an isotope of hydrogen with atomic mass=2. Deuterium can be introduced into organic compounds by using reagents in which hydrogen has been replaced by deuterium. Outline preparation of the following compounds from the same alkene using appropriate deuterium-containing reagents.arrow_forwardWrite a step by step mechanism. Includes resonance structures that exist in the transition states.arrow_forwardUnder strongly acidic conditions, hexane is observed to undergo an isomerization process, during which it is converted into branched alkanes having the same molecular formula as hexane (C6H₁4). The rate at which each constitutional isomer is produced is related to the type of acid that is used in the reaction and the degree of mixing that occurs during the reaction (J. Chem. Soc., Perkins Trans. 2 1999, 12, 2715-2718). Step 1 Draw a bond-line structure of hexane. Draw Your Solution There are four branched isomers of hexane. Draw bond-line structures of all four of its isomers. Draw Your Solutionarrow_forward
- Following is an example of a type of reaction known as a Diels-Alder reaction 1,3-Pentadiene Ethylene 3-Methylcyclohexene (a racemic mixture) The Diels-Alder reaction between a diene and an alkene is quite remarkable in that it is one of the few ways that chemists have to form two new carbon-carbon bonds in a single reaction. Given what you know about the relative strengths of carbon-carbon sigma and pi bonds, would you predict the Diels-Alder reaction to be exothermic or endothermic? Explain your reasoning.arrow_forwardComplete the following answer using the data provided here: • Molecular formula: C5H10O2 • Important IR data (cm-1): 3150-3010 (many); 1740; 1248; 1050 • All 'H NMR data (ppm, splitting, integration): 3.66 ppm (s), 3; 2.20 ppm (t), 2; 1.80 ppm (m), 2; 0.98 ppm (t), 3 Which of the following best fit this spectroscopic data? OH H. A C D Earrow_forwardAlkyl halides undergo nucleophilic substitution and elimination reactions. When the kinetics of the reaction are measured, if the rate of the reaction is found to be dependent only upon the concentration of the alkyl halide the reaction is first order. The substitution reaction is thus termed SN1, and the elimination reaction is termed E1. These reactions are unimolecular and occur in two steps. The first step is rate-limiting and involves the loss of the leaving group to form a carbocation. In the second, fast, step the nucleophile adds to the carbocation in the SN1 reaction or elimination occurs to give an alkene in the E1 reaction. Because the carbocation is planar, the nucleophile can add to either face and therefore racemization is usually observed although solvent effects can influence this somewhat. E1 elimination follows Zaitsev’s rule and typically yields the most substituted alkene as the major product. Conditions which favor the SN1/E1 pathway include the use of a weak…arrow_forward
- Alkyl halides undergo nucleophilic substitution and elimination reactions. When the kinetics of the reaction are measured, if the rate of the reaction is found to be dependent only upon the concentration of the alkyl halide the reaction is first order. The substitution reaction is thus termed SN1, and the elimination reaction is termed E1. These reactions are unimolecular and occur in two steps. The first step is rate-limiting and involves the loss of the leaving group to form a carbocation. In the second, fast, step the nucleophile adds to the carbocation in the SN1 reaction or elimination occurs to give an alkene in the E1 reaction. Because the carbocation is planar, the nucleophile can add to either face and therefore racemization is usually observed although solvent effects can influence this somewhat. E1 elimination follows Zaitsev’s rule and typically yields the most substituted alkene as the major product. Conditions which favor the SN1/E1 pathway include the use of a weak…arrow_forwardExplain the substituent effect on properties of selected molecules such as resonance stability, acidity, acidic properties, basic properties and carbocation stabilityarrow_forwardBriefly explain or give an introduction to this. Do an introduction (in a paragraph) Structure Elucidation of Compounds using Several Spectroscopic Techniques. 1H and 13C NMR Spectroscopy IR Spectroscopy Mass Spectroscopyarrow_forward
- (a) How the thermodynamic parameters explain the stability of an organometallic compound? Explain with proper expressions.arrow_forward(3) HI does not undergo free radical addition with 1-butene, even in the presence of a peroxide. Please provide an explanation by analyzing the propagation steps if the same radical reaction occurred with HI. Besides the bond dissociation enthalpies in the appendix, the following approximate bond dissociate enthalpies might be useful to you: ~310 kJ/mol The component of C=C CH3CH₂CH₂CH₂-Br CH3CH₂CH₂CH2-I -290 kJ/mol (C-Br bond) ~235 kJ/mol (C-I bond)arrow_forwardWhen ethylbromide reacts with potassium cyanide in methanol, the major product is CH3CH₂CN. Some CH3CH₂NC is formed as well, however. Write Lewis structures for the cyanide ion and for both products and provide a mechanistic (structures, no text!) explanation for the course of reaction.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
