
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
8th Edition
ISBN: 9780134015187
Author: John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 14, Problem 14.71CP
Interpretation Introduction
Interpretation:
The acid structure should be identified when the ethanol undergoes oxidation.
Concept introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
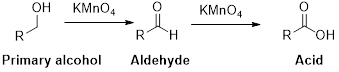
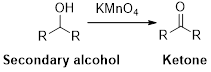
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The Case of the Poisonous Pill: The Poison
By this time, Tylenol has been pulled off the shelves at all of the stores in the area and a
nationwide recall has been announced. Several suspicious capsules have been found and
tested.
The contents of the capsules contain a compound with:
1) A nitrogen content of greater than 15%
2) Carbon content of less than 50%.
What is the poison in the capsule? Write the name of the compound here:
Submit
Soy sauce is made by fermenting a salty combination of soybeans and wheat for 8 to 12 months with a variety of microorganisms, including yeast. After the particles are removed, the resultant sauce is high in lactate and ethanol. What is the process for making these two compounds? To avoid a pronounced vinegary flavor in the soy sauce, air must be kept out of the fermenting tank (vinegar is dilute acetic acid). Why?
3).
reactions of the following alkyl iodide. Danny uses potassium tert-butoxide to promote the reaction
while Claire uses water and high temperatures. One of their reactions has provided a single
elimination isomer while the other has provided a mixture of elimination isomers.
Organic chemistry students Danny and Claire are both performing elimination
Me
H20, heat
Danny's
Reaction
Claire's
Reaction
H.
Me
a) Which student's reaction (Danny or Claire) has provided a single elimination isomer?
b) Draw all the elimination isomers obtained from both students' reactions.
c) Which of the dienes drawn (by you) in question 3b is the most stable? Circle that diene
above.
Chapter 14 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
Ch. 14.1 - Identify each of the following compounds as an...Ch. 14.1 - Prob. 14.2PCh. 14.2 - Prob. 14.3PCh. 14.2 - Prob. 14.4PCh. 14.2 - Prob. 14.5PCh. 14.3 - Prob. 14.6PCh. 14.3 - For each of the following molecules, (i) redraw...Ch. 14.4 - Prob. 14.1MRPCh. 14.4 - Provide the mechanism for the dehydration of...Ch. 14.4 - Prob. 14.3MRP
Ch. 14.4 - Prob. 14.8PCh. 14.4 - What alcohols yield the following alkenes as the...Ch. 14.4 - Prob. 14.10KCPCh. 14.4 - What products would you expect from oxidation of...Ch. 14.4 - Prob. 14.12PCh. 14.4 - Prob. 14.13KCPCh. 14.5 - Prob. 14.14PCh. 14.5 - Prob. 14.15PCh. 14.7 - Prob. 14.1CIAPCh. 14.7 - Prob. 14.2CIAPCh. 14.7 - Prob. 14.3CIAPCh. 14.7 - Prob. 14.16PCh. 14.8 - What disulfides would you obtain from oxidation of...Ch. 14.9 - Prob. 14.18PCh. 14.10 - Prob. 14.19PCh. 14.10 - Prob. 14.20PCh. 14.10 - Prob. 14.4CIAPCh. 14.10 - Prob. 14.5CIAPCh. 14.10 - Prob. 14.6CIAPCh. 14.10 - Prob. 14.7CIAPCh. 14 - Prob. 14.21UKCCh. 14 - Prob. 14.22UKCCh. 14 - Prob. 14.23UKCCh. 14 - Prob. 14.24UKCCh. 14 - Prob. 14.25UKCCh. 14 - How do alcohols, ethers, and phenols differ...Ch. 14 - What is the structural difference between primary,...Ch. 14 - Prob. 14.28APCh. 14 - Prob. 14.29APCh. 14 - The Taxane nucleus is shown here; it is the basis...Ch. 14 - Vitamin E has the structure shown. Identify the...Ch. 14 - Give systematic names for the following alcohols:...Ch. 14 - Give systematic names for the following compound...Ch. 14 - Draw structures corresponding to the following...Ch. 14 - Draw structures corresponding to the following...Ch. 14 - Prob. 14.36APCh. 14 - Locate the alcohol functional groups in the taxane...Ch. 14 - Prob. 14.38APCh. 14 - Prob. 14.39APCh. 14 - Prob. 14.40APCh. 14 - Prob. 14.41APCh. 14 - Prob. 14.42APCh. 14 - Prob. 14.43APCh. 14 - Assume that you have samples of the following two...Ch. 14 - Which of the following alcohols can undergo...Ch. 14 - The following alkenes can be prepared by...Ch. 14 - Prob. 14.47APCh. 14 - Prob. 14.48APCh. 14 - What alcohols would you oxidize to obtain the...Ch. 14 - Prob. 14.50APCh. 14 - What is the structural relationship between a...Ch. 14 - Prob. 14.52APCh. 14 - Prob. 14.53APCh. 14 - Prob. 14.54APCh. 14 - Prob. 14.55APCh. 14 - Prob. 14.56APCh. 14 - Prob. 14.57APCh. 14 - Identify the chiral center(s) in each of the...Ch. 14 - Are the following molecules chiral or achiral? If...Ch. 14 - Prob. 14.60CPCh. 14 - Prob. 14.61CPCh. 14 - 1-Propanol is freely soluble in water, 1-butanol...Ch. 14 - Prob. 14.63CPCh. 14 - Prob. 14.64CPCh. 14 - Prob. 14.65CPCh. 14 - Prob. 14.66CPCh. 14 - Prob. 14.67CPCh. 14 - Prob. 14.68CPCh. 14 - Prob. 14.69CPCh. 14 - Prob. 14.70CPCh. 14 - Prob. 14.71CPCh. 14 - Prob. 14.72CPCh. 14 - (a)Draw all possible cyclic C7H14O alcohol isomers...Ch. 14 - Prob. 14.74GPCh. 14 - Prob. 14.75GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- The structure of a metalloenzyme active site is down below(black picture). Describe, from a chemical and structural perspective, how the reactive site is designed to facilitate its catalytic reaction. The example below(white pitcure) suggests the level of detail that is required. Make sure that you explain what the metal is doing, what the reaction is, and its biological significance.arrow_forwardNumerous research has been conducted to determine how the sequence and structural characteristics of thermophile proteins contribute to their increased stability. Numerous research groups have conducted statistical comparisons between heat-labile and thermostable proteins.(b) What should a protein scientist do if they realized that an enzyme is sensitive to heat?arrow_forwardH,N. NH3 HO, H,O H,N. NH2 COOH СООН glutamic acid glutamine why does the reaction not proceed?arrow_forward
- The biosynthesis of vitamin D₂ is a two-step reaction, requiring two types of concerted pericyclic reactions. Draw in the arrows for each step and identify the structure of precalciferol. If an electrocyclic reaction is required, identify if it occurs through a conrotatory or disrotatory reaction. HO H H ergosterol hv precalciferol Cast HO ergocalciferolarrow_forwardBalance the chemical equation for the oxidation of α-linolenic acid to ATP, CO2, and water. (Use the lowest possible ratio of reactants and products.) +arrow_forwardThe structure of a metalloenzyme active site is down below(black picture). Describe, from a chemical and structural perspective, how the reactive site is designed to facilitate its catalytic reaction. The example below suggests the level of detail that is required. Make sure that you explain what the metal is doing, what the reaction is, and its biological significance.arrow_forward
- Iodine values change during oil hydrogenation. Why these values change during hydrogenation and why would one wish to hydrogenate edible oils?arrow_forwardConsider the reaction: H O C—C—C—SCOA H₂C(CH₂)C= CO H H₂C-(CH₂) What kind of reaction is being performed here? b. What enzyme performs this reaction? OH H O O=C -C—C—C—SCOA H H c. What cofactors, if any, are required for this reaction?arrow_forwarda. What is the name of metabolite 1? b. What is the name of metabolite 2? c. What kind of reaction occurred when 1 was converted to 2? d. What general kind of enzyme might you expect to perform this reaction? e. What cofactor, if any, would be required for this reaction?arrow_forward
- Ethyl alcohol is sometimes used in hospital emergency rooms to treat patients who have ingested radiator or gas-line antifreeze. A major chemical component of these products is methanol. The methanol itself is not especially harmful; however, it is transformed by enzymes called dehydrogenases to the very toxic substances, formaldehyde, and formic acid. Can you explain the biochemical principles behind this medical treatment?arrow_forwardIdentify any combination reactions. (Select all that apply.) C4H10 + O2 → CO2 + H2O S8 + O2 → SO3 NaNO3 → NaNO2 + O2 HgO → Hg + O2 Ga + H2SO4 → Ga2(SO4)3 + H2 AgCl2 + H2 → Ag + HCl none of the above Identify any decomposition reactions. (Select all that apply.) C4H10 + O2 → CO2 + H2O S8 + O2 → SO3 NaNO3 → NaNO2 + O2 HgO → Hg + O2 Ga + H2SO4 → Ga2(SO4)3 + H2 AgCl2 + H2 → Ag + HCl none of the abovearrow_forward1 of 1 3. A sample of glucose reacts in anaerobic respiration. The right- hand box below shows a particle diagram of the moles of substances present after the reaction is complete. fe On a piece of paper draw the "Before" box as shown and draw a particle diagram of the reactant molecules that produced the mixture shown on the right. Key = C2H5OH = CO2 = CgH1206 Sub Before After You will need to draw a diagram to answer this question. On a piece of paper, draw the "Before" box as shown, and then draw a particle diagram of the reactant molecules that produced the mixture shown on the right. Upload an image of your drawing by clicking "Upload files" or by dragging and dropping your file into the box. Or, use your device's camera to take a photo of your work by clicking the camera icon.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license