
Concept explainers
Interpretation:
The solubility should be explained for the propanol is more soluble, whereas ethanethiol and chloroethane is slightly soluble.
Concept introduction:
Solubility of the alcohol in water:
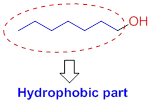
The solubility of the alcohol is mainly depends on the hydrophilic part as well as length of the alkyl chain. If the compound has more number of hydroxyl group or less number of carbon chain which is soluble in water. If the compound has less number of hydroxyl group with more number of carbon chain which is insoluble in water because of hydrophobic part.
The carbon to oxygen ratio of 1:1 to 3:1 is soluble in water (such as methanol, ethanol, and propanol).
Alcohols have two different parts
- (1) Hydrophilic (water-loving),
- (2) Hydrophobic (water-fearing).
Hydrophilic (water-loving):
The compound has the hydroxyl group (

Hydrophobic (water-fearing):
The compound has alkyl chain group is called as hydrophilic.
Hydrogen bond:
The electrostatic attraction between a hydrogen atom and more electronegative atom such as nitrogen (
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Write the formulas of the four singly chlorinated isomers formed when 2-methylbutane reacts with Cl2 in the presence of light.arrow_forwardIn a paragraph form, provide the experimental procedures of the development of the ring structure of α-tetralene by elimination of the water molecule will result in this product being heated in the presence of strong sulphuric acid.arrow_forwardIn solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?arrow_forward
- The reaction of methoxy benzene with hydrogen iodide will yield a phenol and an alkyl halide. Which of following choices is the correct combination of the products?arrow_forwardAn amino acid mixture of phenylalanine, glycine and glutamic acid is to be separated by paper chromatography.The solvent is less polar than water. Which of these amino acids will have the highest Rf value and which the lowest? Explain.arrow_forwardAn amino acid mixture of phenylalanine, glycine and glutamic acid is to be separated by paper chromatography. The solvent is less polar than water. Which of these amino acids will have the highest Rf value and which the lowest? Explain.arrow_forward
- (a) (S)-2-chlorobutane, draw a three-dimensional representation.arrow_forwardPropanamide and methyl acetate have about the same molar mass, both are quite soluble in water, and yet the boiling point of propanamide is 486 K, whereas that of methyl acetate is 330 K. Explain.arrow_forwardPropose a scheme to separate and isolate a mixture containing 4-aminobenzoate, 1,2,4,5-tetrachlorobenzene, and napthalene. Isolate two of the three solids by extraction. (please explain what solvents to use and the process step by step)arrow_forward
- One of these forms of cocaine is relatively insoluble inwater: which form, the free base or the hydrochloride?arrow_forwardIsobutylene, CH2=C(CH3)2, is used to prepare cold-flow rubber. Draw a structure for the addition polymer formed from this alkene.arrow_forwardOne of the main sources of sphingosine in the body is in the cell membrane. What complication could arise from the biological synthesis of ceramide?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





