
Concept explainers
(a)
Interpretation:
The IUPAC name of the given compound is to be assigned.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
Rules for writing the structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ane, ene, yne, ol, al and so on.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
![]()
Answer to Problem 14.5E
The IUPAC name of the given aldehyde is ethanal.
Explanation of Solution
The given compound is shown below.
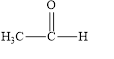
Figure 1
The given compound is aldehyde. The first step in the naming of aldehyde is finding of longest parent chain that contains a carbonyl group. The second step is changing of -e ending of the parent alkane to the suffix -al. The third step is numbering of chain to give the least number to carbonyl carbon, and using the general rules of nomenclature.
The given structure shows the presence of two
The given aldehyde is ethanal.
(b)
Interpretation:
The IUPAC name of the given compound is to be assigned.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
Rules for writing the structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ane, ene, yne, ol, al and so on.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Aldehydes and ketones contain carbonyl ![]() functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
Answer to Problem 14.5E
The IUPAC name of the given aldehyde is
Explanation of Solution
The given compound is shown below.
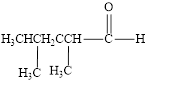
Figure 2
The given compound is aldehyde. The first step in the naming of aldehyde is finding of longest parent chain that contains a carbonyl group. The second step is changing of -e ending of the parent alkane to the suffix -al. The third step is numbering of chain to give the least number to carbonyl carbon, and using the general rules of nomenclature.
The given structure shows the presence of five
The given aldehyde is
(c)
Interpretation:
The IUPAC name of the given compound is to be assigned.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
Rules for writing the structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ane, ene, yne, ol, al and so on.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Aldehydes and ketones contain carbonyl ![]() functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
Answer to Problem 14.5E
The IUPAC name of the given ketone is
Explanation of Solution
The given compound is shown below.
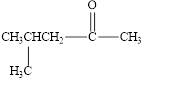
Figure 3
The given compound is ketone. The first step in the naming of ketone is finding of longest parent chain that contains a carbonyl group. The second step is changing of -e ending of the parent alkane to the suffix -one. The third step is numbering of chain to give the least number to carbonyl carbon, and using the general rules of nomenclature.
The given structure shows the presence of five
The given ketone is
(d)
Interpretation:
The IUPAC name of the given compound is to be assigned.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
Rules for writing the structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ane, ene, yne, ol, al and so on.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Aldehydes and ketones contain carbonyl ![]() functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
Answer to Problem 14.5E
The IUPAC name of the given aldehyde is
Explanation of Solution
The given compound is shown below.
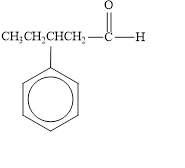
Figure 4
The given compound is aldehyde. The first step in the naming of aldehyde is finding of longest parent chain that contains a carbonyl group. The second step is changing of -e ending of the parent alkane to the suffix -al. The third step is numbering of chain to give the least number to carbonyl carbon, and using the general rules of nomenclature.
The given structure shows the presence of five
The given aldehyde is
(e)
Interpretation:
The IUPAC name of the given compound is to be assigned.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
Rules for writing the structural formula from IUPAC are:
• First identify the word root for the given compound.
• The suffix used in the compound like –ane, ene, yne, ol, al and so on.
• Identify the position, location, and number of the substituent bonded to the carbon chain.
Aldehydes and ketones contain carbonyl ![]() functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
functional group in their parent chain and are named by adding suffix –al and –one to the name of the parent alkane.
Answer to Problem 14.5E
The IUPAC name of the given cyclic ketone is
Explanation of Solution
The given compound is shown below.
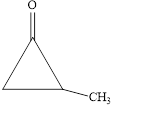
Figure 5
The given compound is cyclic ketone. The first step in the naming of ketone is finding of longest parent chain that contains a carbonyl group. The second step is changing of -e ending of the parent alkane to the suffix -one. The third step is numbering of chain to give the least number to carbonyl carbon, and using the general rules of nomenclature.
The given structure shows the presence of three
The given cyclic ketone is
Want to see more full solutions like this?
Chapter 14 Solutions
Chemistry for Today: General, Organic, and Biochemistry
- Differentiate between single links and multicenter links.arrow_forwardI need help on my practice final, if you could explain how to solve this that would be extremely helpful for my final thursday. Please dumb it down chemistry is not my strong suit. If you could offer strategies as well to make my life easier that would be beneficialarrow_forwardNonearrow_forward
- Definition and classification of boranes.arrow_forwardWhich of the terms explain the relationship between the two compounds? CH2OH Он Он Он Он α-D-galactose anomers enantiomers diastereomers epimers CH2OH ОН O он Он ОН B-D-galactosearrow_forwardHi, I need help on my practice final, If you could offer strategies and dumb it down for me with an explanation on how to solve that would be amazing and beneficial.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





