
Concept explainers
Draw structural formulas for the products of the following hydrolysis reactions:
a. 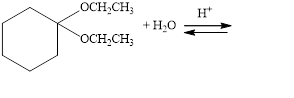
b. 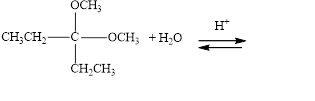
c.
d. 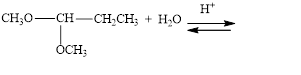
Trending nowThis is a popular solution!

Chapter 14 Solutions
Chemistry for Today: General, Organic, and Biochemistry
- Which of the following is the reaction for the acid hydrolysis of ethyl methanoate? 0 0 1 H-C-0-CH₂-CH3 + H₂O -H-C-OH+CH3-CH₂-OH 0 00 0 0 1 H-C-0-CH3 + H₂0 -H-C-OH+CH₂-OH 0 0 1 0 H-C-0-CH2-CH3 + H20 -H-C-0-CH₂-CH3 1 OH OH 0 0 CH3-C-0-CH3 + H20-CH3-C-OH + CH3-OH CH3-C-ONa+CH-OHarrow_forward16. What is one use for sodium and potassium salts of long-chain carboxylic acids? Oa. food preservative b. athletes foot treatment C. soaps d. pH buffer e. blood anticoagulantarrow_forward1. Write the equation for the hydrolysis of aniline (C6H5NH2), an organic base that smells like rotten fish. 2. Determine whether the following conjugates can undergo hydrolysis. Type either YES or NO. If it can hydrolyze, write the hydrolysis equation. A. ClO4- B. CH3NH3+arrow_forward
- Some coca cola products contain small amounts of (food grade) phosphoric acid. Why might the presence of this acid lead some people to identify coca cola as a helpful remedy for stomach aches?arrow_forwardWhat is the role of phenolphthalein in the neutralization reaction? Draw the structure of phenolphthalein under acidic and basic conditions.arrow_forwardHO. OH NH2 Reactant A Reactant B A "Hydrazone"arrow_forward
- Indicate the product(s) of the reaction: Draw Your Solution Ethyl 2-Methylbutanoate Responsible for the smell of ripe apples 1) LIAIH4 2) H3O* ?arrow_forwardLactic acid, CH3CH(OH)COOH, is a weak monoprotic acid with a melting point of 53 C. It exists as two enantiomers (Sec. 7-2f) that have slightly different Ka values. The D form has a Ka of 1.5 104 and the L form has a Ka of 1.6 104. The D form is synthesized by some bacteria. The L form is produced in muscle cells during anaerobic metabolism in which glucose molecules are broken down into lactic acid and molecules of adenosine triphosphate (ATP) are formed. When lactic acid builds up too rapidly in muscle tissue, severe pain results. (a) Which form of lactic acid (D or L) is the stronger acid? Explain your answer. (b) Determine the pKa that would be measured for a 50:50 mixture of the two forms of lactic acid in aqueous solution, pKa = log Ka (c) A solution of D-lactic acid is prepared. Use HL as a general formula for lactic acid, and write the equation for the ionization of lactic acid in water. (d) If 0.100-M solutions of these two acids (D and L) were prepared, calculate what the pH of each solution would be. (e) Before any lactic acid dissolves in the water, what reaction determines the pH? (f) Calculate the pH of a solution made by dissolving 4.46 g D-lactic acid in 500. mL of water. (g) Calculate the volume (mL) of 1.15-M NaOH(aq) required to completely neutralize 4.46 g of pure lactic acid. (h) Calculate the pH of the solution when exactly enough NaOH was added to neutralize all of the lactic acid for (i) the D form; (ii) the L form; and (iii) a 50:50 mixture of the two forms.arrow_forwardRank the following substances in order of increasing acidity: (a) (CH3)2CHOH, HC≡CH, (CF3)2CHOH, CH3OH (b) Phenol, p-methylphenol, p-[trifluoromethyl) phenol (c) Benzyl alcohol, phenol, p-hydroxybenzonic acidarrow_forward
- What acid is responsible for the sour taste of lemons, limes, and oranges?arrow_forwardWhich of the following common food additives gives a basic solution when dissolved in water? (a) NaNO3 (used as a meat preservative) (b) NaC6H5CO2(Sodium benzoate, used as a soft drink preservative) (c) NaH2PO4(used as a emulsifier in the manufacture of pasteurized cheese)arrow_forwardGive the product of the following reaction: a 'COOH COOH b COOH COOH KMnO4 но C COOH XX. COOH darrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





