
Essential Organic Chemistry, Global Edition
3rd Edition
ISBN: 9781292089034
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 45P
Describe how the following compounds can be synthesized using reagents that contain no more than three carbons:
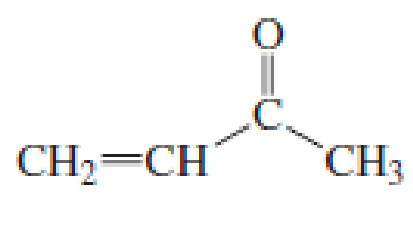
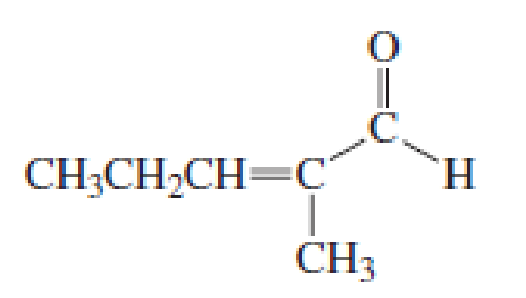
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What product is formed when each compound is treated with K 2Cr 2O 7? With some compounds, no reaction occurs.
Explain how 1-butanol can be converted into the following compounds:
Describe how the following compound can be synthesized from compounds containing no more than six carbons
Chapter 13 Solutions
Essential Organic Chemistry, Global Edition
Ch. 13.1 - Identify the most acidic hydrogen in each...Ch. 13.1 - Prob. 2PCh. 13.1 - Prob. 3PCh. 13.1 - Prob. 4PCh. 13.1 - Explain why HO cannot remove a proton from the...Ch. 13.2 - Prob. 6PCh. 13.2 - Prob. 7PCh. 13.3 - Prob. 8PCh. 13.3 - Prob. 9PCh. 13.3 - Prob. 10P
Ch. 13.4 - Prob. 11PCh. 13.5 - Prob. 12PCh. 13.5 - Prob. 13PCh. 13.6 - Prob. 14PCh. 13.7 - Prob. 16PCh. 13.8 - Prob. 17PCh. 13.8 - Prob. 18PCh. 13.8 - Prob. 19PCh. 13.9 - Prob. 20PCh. 13.10 - Propose a mechanism for the formation of...Ch. 13.10 - Prob. 22PCh. 13.10 - a. If the biosynthesis of palmitic acid were...Ch. 13 - Draw the enol tautomers for each of the following...Ch. 13 - Number the following compounds in order from...Ch. 13 - Prob. 26PCh. 13 - Explain why the pKa of a hydrogen bonded to the...Ch. 13 - Prob. 28PCh. 13 - Prob. 29PCh. 13 - Prob. 30PCh. 13 - Prob. 31PCh. 13 - Prob. 32PCh. 13 - Prob. 33PCh. 13 - Using cyclopentanone as the reactant, show the...Ch. 13 - Prob. 35PCh. 13 - Prob. 36PCh. 13 - Prob. 37PCh. 13 - Prob. 38PCh. 13 - Prob. 39PCh. 13 - Prob. 40PCh. 13 - Prob. 41PCh. 13 - Prob. 42PCh. 13 - Prob. 43PCh. 13 - Prob. 44PCh. 13 - Describe how the following compounds can be...Ch. 13 - Prob. 46PCh. 13 - Which would require a higher temperature:...Ch. 13 - Prob. 48PCh. 13 - Propose a mechanism for the following reaction:Ch. 13 - Show how the following compounds could be...Ch. 13 - Prob. 51PCh. 13 - Prob. 52P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Describe how 1-ethylcyclohexanol can be prepared from cyclohexane. You can use any inorganic reagents, any solvents, and any organic reagents as long as they contain no more than two carbons.arrow_forwardUsing the given starting material, any necessary inorganic reagents and catalysts, and any carbon-containing compounds with no more than two carbons, indicate how each of the following compounds can be prepared:arrow_forwardUsing the given starting material, any necessary inorganic reagents and catalysts, and any carbon-containing compounds with no more than three carbons, indicate how each of the following compounds can be prepared:arrow_forward
- Name the following compound; CH3 CH₂CHCHCH₂CH₂CH₂CHCH₂CH3 O 3-Methyl-8-propylnon-1-ene O 7-Ethyl-3-methyldec-1-ene O 8-Methyl-2-propylnonane O 8-Methyl-2-propylnon-1-ene CH₂CH₂CH3arrow_forwardUsing the given starting material and any necessary organic or inorganic reagents, indicate how the desired compounds could be synthesized:arrow_forwardName and draw the structure of the major product in the following reactions of cyclohexanone: A) Reduction with NaBH4 in ethanol, followed by H+ B) Reaction with excess ethanol in the presence of H+arrow_forward
- What alkene is needed to synthesize each 1,2-diol using [1] OsO4 followed by NaHSO3 in H2O; or [2] CH3CO3H followed by −OH in H2O?arrow_forwardSorbitol is a sweetener often used in chewing gum and diet drinks. It is obtained by the following reaction. Which of the following functional group transformations occur during this reaction? СНО CH2OH Н-с-ОН Н-с-ОН Но-с-н Н2 Но-с-н Н-с-ОН Н-с-оН Н-с-ОН н-с-он CH2OH CH-Он glucose sorbitol a. The aldehyde becomes an alcohol. b. An alcohol becomes an aldehyde. c. An alcohol disappears. d. A new aldehyde appears. e. All of the abovearrow_forwardRank the following alcohols in order of increasing ease of acid-catalyzed dehydration. Provide the structure of the dehydration product (alkene) from each alcohol. OH OH 3 1 a OHarrow_forward
- Alcohols undergo dehydration reactions in the presence of an acid catalyst. Which of the following compounds yields only a single alkene product upon dehydration?arrow_forwardHow could the following compounds be prepared, using cyclohexene as a starting material?arrow_forwardAn alkene with the molecular formula C10H18is treated with ozone and then with zinc and acetic acid. The only product isolated from these reactions is: What is the structure of the alkene? I O III OV O IV ΟΙ IV II Varrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY