
Interpretation:
The type of intermolecular forces in the following compound should be determined.
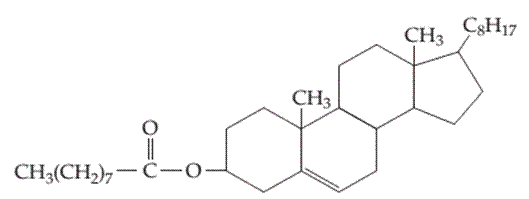
Concept introduction:
Liquid crystals are defined as a phase in which substance exhibits properties of both liquids and solids. Liquid crystal flow like a liquid but their arrangement of the molecule as well as intermolecular forces is like solid.
Liquid crystal molecules are made up of six-membered rings with on terminal polar group, a linkage group and a side chain of carbon atoms. Each carbon atom in liquid crystal molecules has trigonal planar geometry.
The molecules are rigid. The rigidity is increased due to the presence of double-bonded linkage groups such as
The terminal polar groups exhibit strong intermolecular forces such as strong dipole-dipole interaction or dipole−induced dipole interaction and hydrogen bond.
Types of liquid crystal are as follows:
- Nematic Liquid crystal.
- Smectic Liquid crystal.
1. Nematic Liquid crystal: The molecules in the nematic phase are in the same direction and can move around freely very much like that of liquid. In this, the axis is parallel but the ends are not aligned.
2. Smectic Liquid crystal: The molecules in the smectic phase are perpendicular to the plane and are aligned in layers. In these, the long axis is parallel and also their ends are aligned.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
CHEMISTRY-TEXT
- An amorphous solid can sometimes be converted to a crystalline solid by a process called annealing. Annealing consists of heating the substance to a temperature just below the melting point of the crystalline form and then cooling it slowly. Explain why this process helps produce a crystalline solid.arrow_forwardConsider the iodine monochloride molecule, ICI. Because chlorine is more electronegative than iodine, this molecule is a dipole. How would you expect iodine monochloride molecules in the gaseous state to orient themselves with respect to each other as the sample is cooled and the molecules begin to aggregate? Sketch the orientation you would expect.arrow_forwardSilicon carbide, SiC, is a very hard, high-melting solid. What kind of crystal forces account for these properties?arrow_forward
 World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





