
Consider a steam power plant that operates on a regenerative Rankine cycle and has a net power output of 150 MW. Steam enters the turbine at 10 MPa and 500°C and the condenser at 10 kPa. The isentropic efficiency of the turbine is 80 percent, and that of the pumps is 95 percent. Steam is extracted from the turbine at 0.5 MPa to heat the feedwater in an open feedwater heater. Water leaves the feedwater heater as a saturated liquid. Show the cycle on a T-s diagram, and determine (a) the mass flow rate of steam through the boiler and (b) the thermal efficiency of the cycle. Also, determine the exergy destruction associated with the regeneration process. Assume a source temperature of 1300 K and a sink temperature of 303 K.
(a)
The mass flow rate of steam through the boiler.
Answer to Problem 98RP
The mass flow rate of steam through the boiler is
Explanation of Solution
Draw the
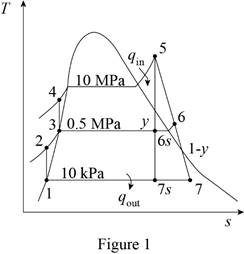
Here, water (steam) is the working fluid of the regenerative Rankine cycle. The cycle involves two pumps.
Write the formula for work done by the pump during process 1-2.
Here, the specific volume is
Write the formula for enthalpy
Write the formula for work done by the pump during process 3-4.
Here, the specific volume is
Write the formula for enthalpy
At state
The steam expanded to the pressure of
The quality of water at state
The enthalpy at state
Here, the enthalpy is
The isentropic efficiency for the process 5-6 is expressed as follows.
At state
The steam enters the condenser at the pressure of
The quality of water at state
The enthalpy at state
Here, the subscript
The isentropic efficiency for the process 5-7 is expressed as follows.
Here, the subscript
Write the formula for heat in
Here, the mass fraction steam extracted from the turbine to the inlet mass of the boiler
Write the general equation of energy balance equation.
Here, the rate of net energy inlet is
At steady state the rate of change of net energy of the system
Refer Equation (XIII).
Write the energy balance equation for open feed water heater.
Rewrite the Equation (XIV) in terms of mass fraction
Write the formula for net work output of the cycle.
Write the formula for mass flow rate of steam.
Here, the net power output of the cycle is
At state 1: (Pump I inlet)
The water exits the condenser as a saturated liquid at the pressure of
Refer Table A-5, “Saturated water-Pressure table”.
The enthalpy
At state 3: (Pump II inlet)
The water exits the open feed water heater as a saturated liquid at the pressure of
Refer Table A-5, “Saturated water-Pressure table”.
The enthalpy
At state 5:
The steam enters the turbine as superheated vapor.
Refer Table A-6, “Superheated water”.
The enthalpy
Refer Figure 1.
At state
The steam expanded to the pressure of
Refer Table A-5, “Saturated water-Pressure table”.
Obtain the following properties corresponding to the pressure of
At state
The steam enters the condenser at the pressure of
Refer Table A-5, “Saturated water-Pressure table”.
Obtain the following properties corresponding to the pressure of
Conclusion:
Substitute
Substitute
Substitute
Substitute
From Figure 1.
Substitute
Substitute
Equation (VI).
Substitute
Substitute
Substitute
Equation (IX).
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
Thus, the mass flow rate of steam through the boiler is
(b)
The thermal efficiency of the cycle and the exergy destruction associated with the regeneration process.
Answer to Problem 98RP
The thermal efficiency of the cycle is
Explanation of Solution
Write the formula for thermal efficiency of the cycle
Write the formula for exergy destruction associated with the regeneration cycle.
Here, the entropy generation is
Conclusion:
Substitute
Thus, the thermal efficiency of the cycle is
Consider the regeneration process- open feed water heater, process states 6,2,3.
Here,
Substitute
Thus, the exergy destruction associated with the regeneration process is
Want to see more full solutions like this?
Chapter 10 Solutions
Thermodynamics: An Engineering Approach
Additional Engineering Textbook Solutions
DeGarmo's Materials and Processes in Manufacturing
Fundamentals of Heat and Mass Transfer
Fundamentals Of Thermodynamics
Fluid Mechanics: Fundamentals and Applications
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
Vector Mechanics for Engineers: Dynamics
- Consider a steam power plant that operates on a regenerative Rankine cycle and has a net power output of 150 MW. Steam enters the turbine at 10 MPa and 500°C and the condenser at 10 kPa. The isentropic efficiency of the turbine is 80 percent, and that of the pumps is 95 percent. Steam is extracted from the turbine at 0.5 MPa to heat the feedwater in an open feedwater heater. Water leaves the feedwater heater as a saturated liquid. Determine the fractions of steam extracted from the turbine. Turbine Boiler Open FWH Condenser Pump II Pump Iarrow_forwardConsider a steam power plant that operates on a reheat Rankine cycle and has a net power output of 80 MW. Steam enters the high-pressure turbine at 10 MPa and 500°C and the low-pressure turbine at 1.4 MPa and 500°C. Steam leaves the condenser as a saturated liquid at a pressure of 10 kPa. Assume both turbine and compressor are isentropic. Determine the mass flow rate of the steam. The mass flow rate of the steam is 74 x kg/s.arrow_forwardConsider a steam power plant that operates on a simple ideal Rankine cycle and has a net power output of 45MW. Steam enters the turbine at 7 MPa and 500 ˚C and is cooled in the condenser at a pressure of 10kPa by running cooling water from a lake through the tubes of the condenser at a rate of 2000 kg/s. Show the schematic diagram of the power plant and draw the cycle on a T-s diagram with respect to saturation lines. Also, determine (a) the thermal efficiency of the cycle, (b) the mass flow rate of the steam, and (c) the rise in temperature of the cooling water. Repeat your calculations assuming an isentropic efficiency of 85 and 90 for the turbine and the pump, respectively.arrow_forward
- Consider a steam power plant that operates on a reheat Rankine cycle and has a net power output of 80 MW. Steam enters the high-pressure turbine at 10 MPa and 500°C and the low-pressure turbine at 1.8 MPa and 500°C. Steam leaves the condenser as a saturated liquid at a pressure of 10 kPa. Assume both turbine and compressor are isentropic. NOTE: This is a multi-part question. Once an answer is submitted, you will be unable to return to this part. Determine the quality (or temperature, if superheated) of the steam at the turbine exit. The quality of the steam at the turbine exit isarrow_forwardConsider a steam power plant that operates on a reheat Rankine cycle and has a net power output of 80 MW. Steam enters the high-pressure turbine at 10 MPa and 500°C and the low-pressure turbine at 1 MPa and 500°C. Steam leaves the condenser as a saturated liquid at a pressure of 10 kPa. The isentropic efficiency of the turbine is 80%, and that of the pump is 95 %. Show the cycle on a T-s diagram with respect to saturation lines, and determine (a)the quality (or temperature, if superheated) of the steam at the turbine exit, (b)the thermal efficiency of the cycle, and (c)the mass flow rate of the steam.arrow_forwardConsider a steam power plant that operates on a reheat Rankine cycle and has a net power output of 80 MW. Steam enters the high-pressure turbine at 10 MPa and 500°C and the low-pressure turbine at 1.4 MPa and 500°C. Steam leaves the condenser as a saturated liquid at a pressure of 10 kPa. Assume both turbine and compressor are isentropic. Determine the thermal efficiency of the cycle. Use steam tables. The thermal efficiency of the cycle is 38.55 %.arrow_forward
- Consider a steam power plant that operates on a reheat Rankine cycle and has a net power output of 80 MW. Steam enters the high-pressure turbine at 10 MPa and 500C and the low-pressure turbine at 1 MPa and 500C. Steam leaves the condenser as a saturated liquid at a pressure of 10 kPa. The isentropic efficiency of the turbine is 80 percent, and that of the pump is 95 percent. (a) Determine the quality (or temperature, if superheated) of the steam at the turbine exit, (b) Determine the thermal efficiency of the cycle, (c) Determine the mass flow rate of the steam, (d) Show the cycle on a T-s diagram with respect to the vapor dome and specifying T and s in all principal points of the cycle.arrow_forwardConsider a steam power plant that operates on the ideal reheat Rankine cycle. The plant maintains the boiler at 17.5 MPa, the reheater at 2 MPa, and the condenser at 50 kPa. The temperature is 550°C at the entrance of the high-pressure turbine, and 300°C at the entrance of the low-pressure turbine. Determine the thermal efficiency of this system. Use steam tables. The thermal efficiency of this system is 74 %.arrow_forwardConsider a steam power plant that operates on a simple ideal Rankine cycle and has a net power output of 120 MW. Steam enters the turbine at 10 MPa and 550°C and is cooled in the condenser at a pressure of 15 kPa by running cooling water from a nearby source the condenser at a rate of 3000 kg/s. The isentropic efficiency of the pump is 95% and that of the turbine is 90%. If h1= 226, h2 = 240k/kg. h3 = 3520 kj/kg, and h4=2210kj/kg. Determine the Actual mass flow rate of steam in kg/sarrow_forward
- Consider an 80 MW coal-fired steam power plant that runs on a reheat Rankine cycle. Steam enters the high-pressure turbine at 10 MPa and 500°C and the low-pressure turbine at 1 MPa and 500°C. Steam leaves the condenser as a saturated liquid at 10 kPa. Determine the volume flow rate of sea water (S.G. = 1.05) used in the condenser, if the allowable temperature rise is 5°C. in cubic meters per hourarrow_forwardThe first steam power plant operates on an ideal reheat Rankine cycle. Steam enters the first turbine at 8 MPa and 500 C and leaves at 3 MPa. Steam is then reheated at a constant pressure to 500 C before it expands to 20 kPa in the low-pressure turbine. Assuming that heat is being added to the boiler from a high temperature source at 1800 K and the condenser is transferring heat to a temperature sink at 300 K. Investigate the effect of changing high and low pressures in the cycle described in scenario 1 on the overall performance (i.e., thermal efficiency). (Hint: First you will have to change the high-pressure value then observe how it will affect the efficiency, then change the low pressure and observe how the efficiency of the cycle will change).arrow_forwardConsider an 80 MW coal-fired steam power plant that runs on a reheat Rankine cycle. Steam enters the high-pressure turbine at 10 MPa and 500°C and the low-pressure turbine at 1 MPa and 500°C. Steam leaves the condenser as a saturated liquid at 10 kPa. Calculate the plant monthly consumption of coal if its heating value is 23,247 kJ/kg and the boiler has an efficiency of 87%. in metric tonsarrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





