
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10.7, Problem 28P
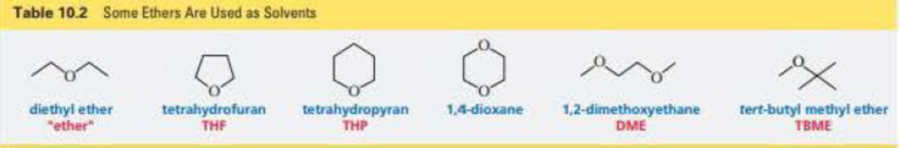
Would you expect the reactivity of a five-membered ring ether such as tetrahydrofuran (Table 10.2) to be more similar to the reactivity of an
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Dehydrohalogenation of 1-chloro-1-methylcyclopropane affords two alkenes (A and B) as products. Explain why A is the major product despite the fact that it contains the less substituted double bond.
Define the below statement ?
Benzene's six π electrons make it electron rich, so it reacts withelectrophiles.
The formate and methoxide ions are both formed by removing a hydrogen atom from a corresponding neutral compound. Given that each process involves the breaking of an O-H bond, explain why the formate ion is so much more stable than the methoxide ion.
Chapter 10 Solutions
Organic Chemistry (8th Edition)
Ch. 10.1 - Why are NH3 and CH3NH2 no longer nucleophiles when...Ch. 10.1 - Explain the difference in reactivity between...Ch. 10.1 - Prob. 5PCh. 10.1 - Prob. 6PCh. 10.1 - Prob. 8PCh. 10.2 - Prob. 9PCh. 10.3 - Prob. 11PCh. 10.3 - Show how 1-propanol can be converted into the...Ch. 10.4 - Which of the following alcohols dehydrates the...Ch. 10.4 - Prob. 14P
Ch. 10.4 - Prob. 15PCh. 10.4 - Propose a mechanism for each of the following...Ch. 10.4 - Draw the product of each of the following...Ch. 10.4 - Explain why the following alcohols, when heated...Ch. 10.4 - What stereoisomers are formed in the following...Ch. 10.4 - Prob. 20PCh. 10.4 - What alcohol would you treat with phosphorus...Ch. 10.5 - Prob. 22PCh. 10.6 - What are the major products obtained when each of...Ch. 10.6 - Prob. 26PCh. 10.7 - Prob. 27PCh. 10.7 - Would you expect the reactivity of a five-membered...Ch. 10.7 - Prob. 29PCh. 10.7 - What products are obtained from the reaction of...Ch. 10.7 - Prob. 31PCh. 10.7 - Prob. 32PCh. 10.7 - Prob. 33PCh. 10.8 - Draw the mechanism for formation of the two...Ch. 10.8 - Prob. 35PCh. 10.8 - Prob. 36PCh. 10.8 - How do the major products obtained from...Ch. 10.8 - Explain why the two arene oxides in Problem 38...Ch. 10.8 - Which compound is more likely to be carcinogenic?Ch. 10.8 - Three arene oxides can be obtained from...Ch. 10.9 - Explain why the half-life (the time it takes for...Ch. 10.10 - Prob. 43PCh. 10.10 - Prob. 44PCh. 10.10 - Prob. 45PCh. 10.10 - Prob. 46PCh. 10.10 - Prob. 47PCh. 10.10 - Describe a synthesis for each of the following...Ch. 10.11 - Using an alkyl halide and a thiol as starting...Ch. 10.11 - The following three nitrogen mustards were studied...Ch. 10.11 - Why is melphalan a good cancer drug?Ch. 10.11 - Prob. 53PCh. 10.12 - Propose a mechanism for the following reaction:Ch. 10 - Prob. 55PCh. 10 - Which compound is more likely to be carcinogenic?Ch. 10 - Prob. 57PCh. 10 - Prob. 58PCh. 10 - When heated with H2SO4, both...Ch. 10 - What is the major product obtained from the...Ch. 10 - Write the appropriate reagent over each arrow.Ch. 10 - What alkenes would you expect to be obtained from...Ch. 10 - Prob. 63PCh. 10 - Prob. 64PCh. 10 - When deuterated phenanthrene oxide undergoes a...Ch. 10 - An unknown alcohol with a molecular formula of...Ch. 10 - Explain why the acid-catalyzed dehydration of an...Ch. 10 - Prob. 68PCh. 10 - Prob. 69PCh. 10 - Propose a mechanism for the following reaction:Ch. 10 - What product would be formed if the four-membered...Ch. 10 - Which of the following ethers would be obtained in...Ch. 10 - Using the given starting material any necessary...Ch. 10 - Prob. 74PCh. 10 - When 3-methyl-2-butanol is heated with...Ch. 10 - Draw structures for compounds AF.Ch. 10 - Propose a mechanism for each of the following...Ch. 10 - How could you synthesize isopropyl propyl ether,...Ch. 10 - When ethyl ether is heated with excess HI for...Ch. 10 - When the following seven-membered ring alcohol is...Ch. 10 - Ethylene oxide reacts readily with HO because of...Ch. 10 - Describe how each of the following compounds could...Ch. 10 - Propose a mechanism for each of the following...Ch. 10 - Triethylene glycol is one of the products obtained...Ch. 10 - Prob. 85PCh. 10 - Propose a mechanism for the following reaction:Ch. 10 - Prob. 87PCh. 10 - An ion with a positively charged nitrogen atom in...Ch. 10 - The following reaction takes place several times...Ch. 10 - Prob. 90PCh. 10 - Propose a mechanism for each of the following...Ch. 10 - A vicinal diol has OH groups on adjacent carbons....Ch. 10 - Prob. 93PCh. 10 - Prob. 94PCh. 10 - Two stereoisomers are obtained from the reaction...Ch. 10 - Propose a mechanism for each or the following...Ch. 10 - Triethylenemelamine (TEM) is an antitumor agent....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Explain why the C=C of an enol is more nucleophilic than the C=C of an alkene, despite the fact that the electronegative oxygen atom of the enol inductively withdraws electron density from the carbon–carbon double bond.arrow_forwardWrite the structures of all the constitutionally isomeric ethers of molecular formula C5H12O, and give an acceptable name for each.arrow_forwardCH3 OCH2CH2CH CH3 (10) (11) alkyl bromide/ phenyl bromide sodium alkoxide/ sodium phenoxidearrow_forward
- Chlordane, like DDT, is an alkyl halide that was used as an insecticide for crops such as corn and citrus and for lawns. In 1983, it was banned for all uses except against termites, and in 1988, it was banned for use against termites as well. Chlordane can be synthesized from two reactants in one step. One of the reactants is hexachlorocyclopentadiene. What is the other reactant?arrow_forwardExplain why heats of hydrogenation cannot be used to determine the relative stability of 2-methylpent-2-ene and 3-methylpent-1-ene. A, B, and C, each subjected to hydrogenation. The number of rings and π bonds refers to the reactant (A, B, or C) prior to hydrogenation.arrow_forward5 AM 7.12. Draw the structural formula of the enol formed in each alkyne hydration reaction; then draw the structural formula of the carbonyl compound with which each enol is in equilibrium. (a) CH, (CH₂), C=CH + H₂O (b) CH, (CH₂),C=CH HgSO4 H₂SO4 1. (sia), BH 2. NaOH/H₂O₂ (an enol) → →(an enol) →arrow_forward
- Cyclopropenones are described as having aromatic character. How would you account for this, given that the ring contains three ℼ-electronarrow_forwardThe structure below is the cyclic ether (epoxide), butene oxide: (1) CH3CH₂ -CH₂ butene oxide How could this compound be prepared from but-1-ene? Explain why butene oxide is much more reactive than its isomer, tetrahydrofuran, which is also a cyclic ether: H₂C-CH₂ H₂C CH₂ tetrahydrofuran Illustrate how butene oxide reacts with ammonia, showing details of the mechanism leading to the final product, C4H11 NO.arrow_forwardAlkyl halides undergo elimination reactions to produce alkenes by the reacting with strong bases as shown in the following reaction (see image). In general, compounds where the halogen is axial (axial position) are much more reactive than those in which they are in the equatorial position. Taking the above into account: a. Which of the following compounds would give a faster elimination reaction: cis-1-bromo-2-tert-butylcyclohexane or trans-1-bromo-2-tert-butylcyclohexane? Draw the corresponding structures and clearly explain the choice.arrow_forward
- Zaitsev's rule for the acid catalyzed dehydration of alcohols states: It states that in a regioselective E1 or E2 reaction the major product is the least stable alkene, i.e., the alkene with the less substituted double bond. O 1) True O 2) Falsearrow_forward4. A certain endothermic reaction X carried out room temperature (273K) has an enthalpy change of 20 Kcal/mol and an entropy change of 10 Kcal/K/mol. i) Calculate the Gibbs free energy (AG) of this reaction. ii) Is this reaction spontaneous or non-spontaneous? Give a reason for your answer 5. Thiomethane reacts with compound A via an SN1 reaction. Complete its reaction equation below. ii) SH Compound A ii) Draw the mechanism of reaction for this reaction. 6. Butyl Chloride undergoes an elimination reaction when reacted with sodium ethoxide to form butene. The reaction equation is given below with two reaction products A and B. low inevloa CI noitsups p *OEt B Which is the major product, and which is minor product. (Hint: Look up how the size of the base affects the elimination reaction product) Give a reason for your answer.arrow_forwardRank compounds 1 – 4 (shown below) in terms of decreasing rates of cyclic ether formation. Use the provided ranking options.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY