
Concept explainers
(a)
Interpretation:
The product of the given reaction should be given, respect with the given conditions.
Concept introduction:
Chromic Acid:
Chromic Acid (
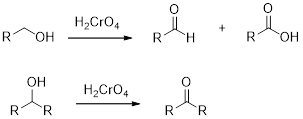
(b)
Interpretation:
The product of the given reaction should be given, respect with the given conditions.
Concept introduction:
Hypochlorous acid
Hypochlorous acid (
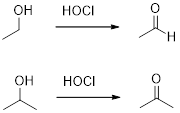
(c)
Interpretation:
The product of the given reaction should be given, respect with the given conditions.
Concept introduction:
Swern oxidation:
Primary and secondary alcohols undergoes oxidation reaction using dimethyl sulfoxide, oxalyl chloride and triethyl
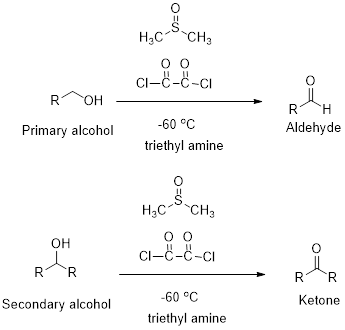
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Organic Chemistry (8th Edition)
- 11. The stabilized ether contains a stadilizer-antioxidant?A) Methyl alcohol in a concentration of not more than 0.1%B) Para-phenylenediamineC) Resorcinol not less than 0.1%D) Thymol not less than 0.1%arrow_forward5. WHAT IS GRIGNARD REACTION? WHAT CONSTITUTES A GRIGNARD REAGENT? 6. WHAT IS KETO-ENOL TAUTOMERIZATION?arrow_forwardWhich alcohol reacts most rapidly with the Lucas reagent?A) benzyl alcoholB) methanolC) 2-propanolD) isobutanolAnswer:arrow_forward
- 1. Explain the solubility behavior of the alcohols and phenols in water as a function of: a. Branching in the structure b. Relative proportion of hydrophilic to hydrophobic bonds 2. Based on oxidation test, classify the alcohols and phenols according to the following categories: easily oxidizable, oxidizable, and resistant to oxidation. 3. Explain in 2 – 3 sentences the reactivity differences of the alcohols towards the Lucas reagent. 4. Explain in 2 – 3 sentences the solubility of alcohols and phenols in water.arrow_forwardDraw the product(s) of the reaction between 1-propyne (1 mole) and the reagents given below. A) 2 moles HBr B) excess H2 over Pd-C C) excess H2 in Lindlar’s catalystarrow_forwardExample of compound with oxidizable (using PCC and H₂CrO₄) carbonyl: A.) p-formylbenzoic acid B.) 2-oxocyclohexanaoic acid C.) Bicyclo[2.2.1]heptan-7-one D.) 1,3-cyclononanedione E.) All of the given choicesarrow_forward
- When synthesizing acetals and ketals, what is the main driving force behind whether the reaction favors the carbonyl species or the acetal/ketal? a the catalytic acid b solvent c temperature d Le Châtelier's Principlearrow_forwardWe have covered several oxidants that use a multi-valent atom (Cr, Cl, S, or I) as their active species, going from a higher oxidation state before the oxidation to a lower oxidation state after oxidizing the alcohol. Draw the structure of the following atoms, before and after the oxidation of an alcohol to a ketone or aldehyde. How many bonds to oxygen does each atom have before and after the oxidation? (a) the I in the DMP reagent (b) the carbinol C in the alcohol that is oxidizedarrow_forwardDo ethers have a good leaving group ?arrow_forward
- What reagent can be used from compound F to G? (NaH / NaOH / LiAlH4+hydronium quench / CrO3 Jones)arrow_forward3 Organic Chemistry, helppp with ALL PARTS(A&B) Consider the monoterpene linalool, a natural product used in the fragrance industry. When linalool (right panel) is treated with a strong protic acid in the presence of heat, 12 distinct products form. a. Given the initial structure of linalool, draw the structure of these 12 products b. Show the structure of the five intermediates that facilitate the formation of these 12 products and use arrow pushing to show how the intermediates formarrow_forwardConvert benzene into attached compound. You may also use any inorganic reagents and organic alcohols having four or fewer carbons. One step of the synthesis must use a Grignard reagent.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





