
(a)
Interpretation:
The appropriate reagents should be given for each steps in the given reaction.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction of alcohol with strong acid like sulfuric acid is known as dehydration reaction.
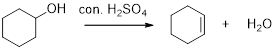
The stability of carbocation is given below, Tertiary carbocation is more stable than the secondary and primary.
The alcohols is reaction with acids like hydrochloric acid or hydrobromic, which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes elimination reaction which yields the corresponding
Hydroboration:
Hydroboration is the addition of a hydrogen-boron bond to the Carbon-Carbon, Carbon-Nitrogen, and Carbon-Oxygen double bonds and Carbon-Carbon triple bonds.
When alkene undergoes hydroboration using alkyl borane and hydrogen peroxide followed by hydrolysis which yields the alcohol. The formation of alcohol is depends on the less hindered carbon of the double bond.
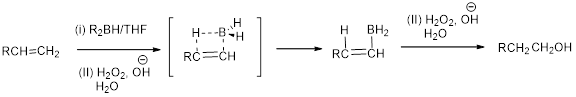
In the nucleophilic substitution reaction, the
In
Reactant and nucleophile are present at the rate determination step.
The order of species involving in
Tertiary < Secondary < Primary
Ozonolysis:
Alkene reacts with ozone to gives
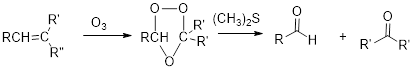
Oxidation of alcohol:
Alcohols reacts with hypochlorous (oxidizing agent) in the presence of acetic acid which yields the corresponding aldehyde and
Primary alcohols gives aldehyde, secondary alcohols gives ketone.
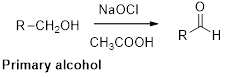
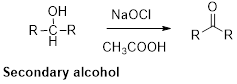
(b)
Interpretation:
The appropriate reagents should be given for each steps in the given reaction.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction of alcohol with strong acid like sulfuric acid is known as dehydration reaction.
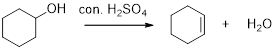
The stability of carbocation is given below, Tertiary carbocation is more stable than the secondary and primary.
The alcohols is reaction with acids like hydrochloric acid or hydrobromic which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes elimination reaction which yields the corresponding alkene as a product.
Hydroboration:
Hydroboration is the addition of a hydrogen-boron bond to the Carbon-Carbon, Carbon-Nitrogen, and Carbon-Oxygen double bonds and Carbon-Carbon triple bonds.
When alkene undergoes hydroboration using alkyl borane and hydrogen peroxide followed by hydrolysis which yields the alcohol. The formation of alcohol is depends on the less hindered carbon of the double bond.

In the nucleophilic substitution reaction, the rate of reaction depends on reactant as well as nucleophile, which are involved in reaction is called bimolecular nucleophilic substitution reaction.
In
Reactant and nucleophile are present at the rate determination step.
The order of species involving in
Tertiary < Secondary < Primary
Ozonolysis:
Alkene reacts with ozone which cleaves the double bond followed by work up with dimethyl sulfide to forms ketone with aldehyde is known as ozonolysis.
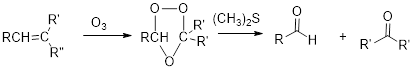
Oxidation of alcohol:
Alcohols reaction with hypochlorous (oxidizing agent) in the presence of acetic acid which yields the corresponding aldehyde and ketones.
Primary alcohols gives aldehyde, secondary alcohols gives ketone.
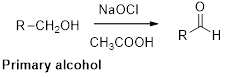
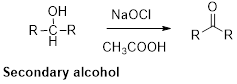
(c)
Interpretation:
The appropriate reagents should be given for each steps in the given reaction.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction of alcohol with strong acid like sulfuric acid is known as dehydration reaction.
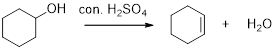
The stability of carbocation is given below, Tertiary carbocation is more stable than the secondary and primary.
The alcohols is reaction with acids like hydrochloric acid or hydrobromic which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes elimination reaction which yields the corresponding alkene as a product.
Hydroboration:
Hydroboration is the addition of a hydrogen-boron bond to the Carbon-Carbon, Carbon-Nitrogen, and Carbon-Oxygen double bonds and Carbon-Carbon triple bonds.
When alkene undergoes hydroboration using alkyl borane and hydrogen peroxide followed by hydrolysis which yields the alcohol. The formation of alcohol is depends on the less hindered carbon of the double bond.

In the nucleophilic substitution reaction, the rate of reaction depends on reactant as well as nucleophile, which are involved in reaction is called bimolecular nucleophilic substitution reaction.
In
Reactant and nucleophile are present at the rate determination step.
The order of species involving in
Tertiary < Secondary < Primary
Ozonolysis:
Alkene reacts with ozone which cleaves the double bond followed by work up with dimethyl sulfide to forms ketone with aldehyde is known as ozonolysis.
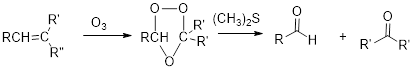
Oxidation of alcohol:
Alcohols reaction with hypochlorous (oxidizing agent) in the presence of acetic acid which yields the corresponding aldehyde and ketones.
Primary alcohols gives aldehyde, secondary alcohols gives ketone.
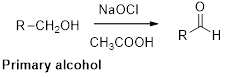
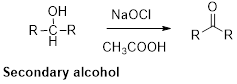
Trending nowThis is a popular solution!

Chapter 10 Solutions
Organic Chemistry (8th Edition)
- Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Select to Edit Arrows H H Select to Add Arrows > H CFCI: Select to Edit Arrows H Select to Edit Arrowsarrow_forwardShow work with explanation needed. don't give Ai generated solutionarrow_forwardShow work. don't give Ai generated solutionarrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

